Unusual cerebral vascular prion protein amyloid distribution in scrapie-infected transgenic mice expressing anchorless prion protein
- PMID: 24252347
- PMCID: PMC3893542
- DOI: 10.1186/2051-5960-1-25
Unusual cerebral vascular prion protein amyloid distribution in scrapie-infected transgenic mice expressing anchorless prion protein
Abstract
Background: In some prion diseases, misfolded aggregated protease-resistant prion protein (PrPres) is found in brain as amyloid, which can cause cerebral amyloid angiopathy. Small diffusible precursors of PrPres amyloid might flow with brain interstitial fluid (ISF), possibly accounting for the perivascular and intravascular distribution of PrPres amyloid. We previously reported that PrPres amyloid in scrapie-infected transgenic mice appeared to delay clearance of microinjected brain ISF tracer molecules.
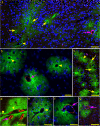
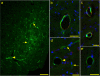
Results: Here we studied distribution of PrPres amyloid on capillaries, arteries and veins to test whether vascular specificity of PrPres corresponded to distribution of ISF tracer molecules. To distinguish PrPres-positive arteries from veins and capillaries, scrapie-infected mouse brains were studied by immunodetection of alpha smooth muscle actin. ISF was studied using fluorescein-labeled ovalbumin microinjected into brain as a tracer. In infected preclinical or clinical mice, PrPres was found mostly on capillaries (73-78%). Lower levels were found on arteries (11-14%) and veins (11-13%). Compared to PrPres, ISF tracer was found at higher levels on capillaries (96-97%), and the remaining tracer was found at a skewed ratio of 4 to 1 on arteries and veins respectively.
Conclusions: PrPres association with blood vessels suggested that ISF flow might transport diffusible PrPres precursor molecules to perivascular sites. However, the different vascular specificity of PrPres and ISF tracer indicated that ISF flow did not alone control PrPres dissemination. Possibly blood vessel basement membrane (BM) components, such as glucosaminoglycans, might concentrate small PrPres aggregates and serve as scaffolds for PrP conversion on multiple vessel types.
Figures

References
-
- Budka H, Aguzzi A, Brown P, Brucher JM, Bugiani O, Gullotta F, Haltia M, Hauw JJ, Ironside JW, Jellinger K. et al. Neuropathological diagnostic criteria for Creutzfeldt-Jakob disease (CJD) and other human spongiform encephalopathies (prion diseases) Brain Pathol. 1995;1(4):459–466. doi: 10.1111/j.1750-3639.1995.tb00625.x. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

