Parasite epigenetics and immune evasion: lessons from budding yeast
- PMID: 24252437
- PMCID: PMC3843538
- DOI: 10.1186/1756-8935-6-40
Parasite epigenetics and immune evasion: lessons from budding yeast
Abstract
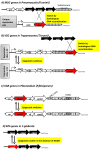
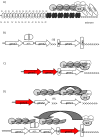
The remarkable ability of many parasites to evade host immunity is the key to their success and pervasiveness. The immune evasion is directly linked to the silencing of the members of extended families of genes that encode for major parasite antigens. At any time only one of these genes is active. Infrequent switches to other members of the gene family help the parasites elude the immune system and cause prolonged maladies. For most pathogens, the detailed mechanisms of gene silencing and switching are poorly understood. On the other hand, studies in the budding yeast Saccharomyces cerevisiae have revealed similar mechanisms of gene repression and switching and have provided significant insights into the molecular basis of these phenomena. This information is becoming increasingly relevant to the genetics of the parasites. Here we summarize recent advances in parasite epigenetics and emphasize the similarities between S. cerevisiae and pathogens such as Plasmodium, Trypanosoma, Candida, and Pneumocystis. We also outline current challenges in the control and the treatment of the diseases caused by these parasites and link them to epigenetics and the wealth of knowledge acquired from budding yeast.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

