Uptake of inorganic mercury by human locus ceruleus and corticomotor neurons: implications for amyotrophic lateral sclerosis
- PMID: 24252585
- PMCID: PMC3893560
- DOI: 10.1186/2051-5960-1-13
Uptake of inorganic mercury by human locus ceruleus and corticomotor neurons: implications for amyotrophic lateral sclerosis
Abstract
Background: Environmental toxins are suspected to play a role in the pathogenesis of amyotrophic lateral sclerosis (ALS). In an attempt to determine which pathways these toxins can use to enter motor neurons we compared the distribution of mercury in the CNS of a human and of mice that had been exposed to inorganic mercury.
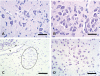
Results: In the human who had been exposed to metallic mercury, mercury was seen predominantly in the locus ceruleus and corticomotor neurons, as well as in scattered glial cells. In mice that had been exposed to mercury vapor or mercuric chloride, mercury was present in lower motor neurons in the spinal cord and brain stem.
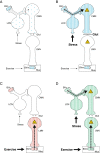
Conclusions: In humans, inorganic mercury can be taken up predominantly by corticomotor neurons, possibly when the locus ceruleus is upregulated by stress. This toxin uptake into corticomotor neurons is in accord with the hypothesis that ALS originates in these upper motor neurons. In mice, inorganic mercury is taken up predominantly by lower motor neurons. The routes toxins use to enter motor neurons depends on the nature of the toxin, the duration of exposure, and possibly the amount of stress (for upper motor neuron uptake) and exercise (for lower motor neuron uptake) at the time of toxin exposure.
Figures


References
-
- Chio A, Calvo A, Mazzini L, Cantello R, Mora G, Moglia C, Corrado L, D’Alfonso S, Majounie E, Renton A, Pisano F, Ossola I, Brunetti M, Traynor BJ, Restagno G. Extensive genetics of ALS: a population-based study in italy. Neurology. 2012;1:1983–1989. doi: 10.1212/WNL.0b013e3182735d36. - DOI - PMC - PubMed
-
- Conte A, Lattante S, Luigetti M, Del Grande A, Romano A, Marcaccio A, Marangi G, Rossini PM, Neri G, Zollino M, Sabatelli M. Classification of familial amyotrophic lateral sclerosis by family history: effects on frequency of genes mutation. J Neurol Neurosurg Psychiatry. 2012;1:1201–1203. doi: 10.1136/jnnp-2012-302897. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

