The operation of two decarboxylases, transamination, and partitioning of C4 metabolic processes between mesophyll and bundle sheath cells allows light capture to be balanced for the maize C4 pathway
- PMID: 24254314
- PMCID: PMC3875822
- DOI: 10.1104/pp.113.228221
The operation of two decarboxylases, transamination, and partitioning of C4 metabolic processes between mesophyll and bundle sheath cells allows light capture to be balanced for the maize C4 pathway
Abstract
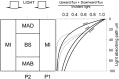
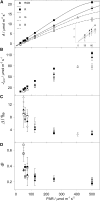
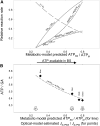
The C4 photosynthesis carbon-concentrating mechanism in maize (Zea mays) has two CO2 delivery pathways to the bundle sheath (BS; via malate or aspartate), and rates of phosphoglyceric acid reduction, starch synthesis, and phosphoenolpyruvate regeneration also vary between BS and mesophyll (M) cells. The theoretical partitioning of ATP supply between M and BS cells was derived for these metabolic activities from simulated profiles of light penetration across a leaf, with a potential 3-fold difference in the fraction of ATP produced in the BS relative to M (from 0.29 to 0.96). A steady-state metabolic model was tested using varying light quality to differentially stimulate M or BS photosystems. CO2 uptake, ATP production rate (JATP; derived with a low oxygen/chlorophyll fluorescence method), and carbon isotope discrimination were measured on plants under a low light intensity, which is considered to affect C4 operating efficiency. The light quality treatments did not change the empirical ATP cost of gross CO2 assimilation (JATP/GA). Using the metabolic model, measured JATP/GA was compared with the predicted ATP demand as metabolic functions were varied between M and BS. Transamination and the two decarboxylase systems (NADP-malic enzyme and phosphoenolpyruvate carboxykinase) were critical for matching ATP and reduced NADP demand in BS and M when light capture was varied under contrasting light qualities.
Figures
References
-
- Allen WA, Richardson AJ. (1968) Interaction of light with a plant canopy. J Opt Soc Am 58: 1023–1028
-
- Baker NR, Long SP (1988) Photosynthesis and temperature, with particular reference to effects on quantum yield. In SP Long, FI Woodward, eds, Plants and Temperature: Society for Experimental Biology Symposium No XXXXII. Company of Biologists, London, pp 347–375 - PubMed
-
- Bellasio C, Griffiths H. (October 8, 2013) Acclimation to low light by C4 maize: implications for bundle sheath leakiness. Plant Cell Environ http://dx.doi.org/10.1111/pce.12194 - PubMed
-
- Berry JA, Farquhar GD (1978) The CO2 concentrating function of C4 photosynthesis: a biochemical model. In D Hall, J Coombs, T Goodwin, eds, Proceedings of the 4th International Congress on Photosynthesis. Biochemical Society, London, pp 119–131
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

