Characterization of VRC01, a potent and broadly neutralizing anti-HIV mAb, produced in transiently and stably transformed tobacco
- PMID: 24256218
- PMCID: PMC4112721
- DOI: 10.1111/pbi.12137
Characterization of VRC01, a potent and broadly neutralizing anti-HIV mAb, produced in transiently and stably transformed tobacco
Abstract
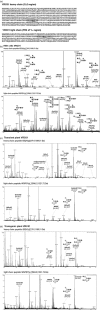
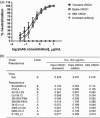
The proposed clinical trial in Africa of VRC01, a potent broadly neutralizing antibody (bNAb) capable of neutralizing 91% of known HIV-1 isolates, raises concerns about testing a treatment which will be too expensive to be accessible by the most important target population, the poor in under-developed regions such as sub-Saharan Africa. Here, we report the expression of VRC01 in plants as an economic alternative to conventional mammalian-cell-based production platforms. The heavy and light chain genes of VRC01 were cloned onto a single vector, pTRAk.2, which was transformed into Nicotiana benthamiana or Nicotiana tabacum using transient and stable expression production systems respectively. VRC01 has been successfully expressed transiently in plants with expression level of approximately 80 mg antibody/kg; stable transgenic lines expressing up to 100 mg antibody/kg were also obtained. Plant-produced VRC01 from both systems showed a largely homogeneous N-glycosylation profile with a single dominant glycoform. The binding kinetics to gp120 IIIB (approximately 1 nM), neutralization of HIV-1 BaL or a panel of 10 VRC01-sensitive HIV-1 Env pseudoviruses of VRC01 produced in transient and stable plants were also consistent with VRC01 from HEK cells.
Keywords: HIV-1 broadly neutralizing antibody; VRC01; pTRAk.2; stable transgenics; transient expression.
© 2013 The Authors. Plant Biotechnology Journal published by Society for Experimental Biology, The Association of Applied Biologists and John Wiley & Sons Ltd.
Figures
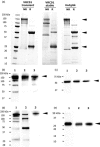

 ), stable transgenic plant ‐(
), stable transgenic plant ‐( ) and
) and  )produced
)produced  ). Error bars denotes ±
). Error bars denotes ± Similar articles
-
Efficient single tobamoviral vector-based bioproduction of broadly neutralizing anti-HIV-1 monoclonal antibody VRC01 in Nicotiana benthamiana plants and utility of VRC01 in combination microbicides.Antimicrob Agents Chemother. 2013 May;57(5):2076-86. doi: 10.1128/AAC.02588-12. Epub 2013 Feb 12. Antimicrob Agents Chemother. 2013. PMID: 23403432 Free PMC article.
-
Rapid high-level production of functional HIV broadly neutralizing monoclonal antibodies in transient plant expression systems.PLoS One. 2013;8(3):e58724. doi: 10.1371/journal.pone.0058724. Epub 2013 Mar 22. PLoS One. 2013. PMID: 23533588 Free PMC article.
-
Prediction of VRC01 neutralization sensitivity by HIV-1 gp160 sequence features.PLoS Comput Biol. 2019 Apr 1;15(4):e1006952. doi: 10.1371/journal.pcbi.1006952. eCollection 2019 Apr. PLoS Comput Biol. 2019. PMID: 30933973 Free PMC article.
-
HIV Broadly Neutralizing Antibodies: VRC01 and Beyond.Adv Exp Med Biol. 2018;1075:53-72. doi: 10.1007/978-981-13-0484-2_3. Adv Exp Med Biol. 2018. PMID: 30030789 Review.
-
Plant made anti-HIV microbicides--a field of opportunity.Biotechnol Adv. 2012 Nov-Dec;30(6):1614-26. doi: 10.1016/j.biotechadv.2012.06.002. Epub 2012 Jun 28. Biotechnol Adv. 2012. PMID: 22750509 Free PMC article. Review.
Cited by
-
Production of Recombinant Proteins in Transgenic Tobacco Plants.Methods Mol Biol. 2022;2480:17-48. doi: 10.1007/978-1-0716-2241-4_2. Methods Mol Biol. 2022. PMID: 35616855
-
Engineering the N-glycosylation pathway of Nicotiana tabacum for molecular pharming using CRISPR/Cas9.Front Plant Sci. 2022 Sep 8;13:1003065. doi: 10.3389/fpls.2022.1003065. eCollection 2022. Front Plant Sci. 2022. PMID: 36161010 Free PMC article.
-
Transient plant production of Salmonella Typhimurium diagnostic antibodies.Biotechnol Rep (Amst). 2019 Feb 12;21:e00314. doi: 10.1016/j.btre.2019.e00314. eCollection 2019 Mar. Biotechnol Rep (Amst). 2019. PMID: 30847285 Free PMC article.
-
A plant-biotechnology approach for producing highly potent anti-HIV antibodies for antiretroviral therapy consideration.J Genet Eng Biotechnol. 2021 Dec 8;19(1):180. doi: 10.1186/s43141-021-00279-z. J Genet Eng Biotechnol. 2021. PMID: 34878628 Free PMC article.
-
Three unrelated protease inhibitors enhance accumulation of pharmaceutical recombinant proteins in Nicotiana benthamiana.Plant Biotechnol J. 2018 Oct;16(10):1797-1810. doi: 10.1111/pbi.12916. Epub 2018 May 24. Plant Biotechnol J. 2018. PMID: 29509983 Free PMC article.
References
-
- Barbaro, G. (2006) Metabolic and cardiovascular complications of highly active antiretroviral therapy for HIV infection. Curr. HIV Res. 4, 79–85 - PubMed
-
- Barbaro, G., Scozzafava, A., Mastrolorenzo, A. and Supuran, C.T. (2005) Highly active antiretroviral therapy: current state of the art, new agents and their pharmacological interactions useful for improving therapeutic outcome. Curr. Pharm. Des. 11, 1805–1843 - PubMed
-
- Bartlett, J.G. (2006) Ten Years of HAART: Foundation for the Future. http://www.medscape.org/viewarticle/523119 (accessed 03 April 2013).
-
- Batard, Y., Hehn, A., Nedelkina, S., Schalk, M., Pallett, K., Schaller, H. and Werck‐Reichhart, D. (2000) Increasing expression of P450 and P450‐reductase proteins from monocots in heterologous systems. Arch. Biochem. Biophys. 379, 161–169 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

