Plastid-bearing sea slugs fix CO2 in the light but do not require photosynthesis to survive
- PMID: 24258718
- PMCID: PMC3843837
- DOI: 10.1098/rspb.2013.2493
Plastid-bearing sea slugs fix CO2 in the light but do not require photosynthesis to survive
Abstract
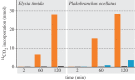
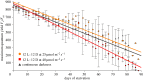
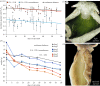
Several sacoglossan sea slugs (Plakobranchoidea) feed upon plastids of large unicellular algae. Four species--called long-term retention (LtR) species--are known to sequester ingested plastids within specialized cells of the digestive gland. There, the stolen plastids (kleptoplasts) remain photosynthetically active for several months, during which time LtR species can survive without additional food uptake. Kleptoplast longevity has long been puzzling, because the slugs do not sequester algal nuclei that could support photosystem maintenance. It is widely assumed that the slugs survive starvation by means of kleptoplast photosynthesis, yet direct evidence to support that view is lacking. We show that two LtR plakobranchids, Elysia timida and Plakobranchus ocellatus, incorporate (14)CO2 into acid-stable products 60- and 64-fold more rapidly in the light than in the dark, respectively. Despite this light-dependent CO2 fixation ability, light is, surprisingly, not essential for the slugs to survive starvation. LtR animals survived several months of starvation (i) in complete darkness and (ii) in the light in the presence of the photosynthesis inhibitor monolinuron, all while not losing weight faster than the control animals. Contrary to current views, sacoglossan kleptoplasts seem to be slowly digested food reserves, not a source of solar power.
Keywords: Elysia; Gastropoda; Kleptoplasty; Sacoglossa; photoautotroph; photosynthetic slugs.
Figures

Similar articles
-
Transcriptomic evidence that longevity of acquired plastids in the photosynthetic slugs Elysia timida and Plakobranchus ocellatus does not entail lateral transfer of algal nuclear genes.Mol Biol Evol. 2011 Jan;28(1):699-706. doi: 10.1093/molbev/msq239. Epub 2010 Sep 9. Mol Biol Evol. 2011. PMID: 20829345 Free PMC article.
-
Comparison of sister species identifies factors underpinning plastid compatibility in green sea slugs.Proc Biol Sci. 2015 Mar 7;282(1802):20142519. doi: 10.1098/rspb.2014.2519. Epub 2015 Feb 4. Proc Biol Sci. 2015. PMID: 25652835 Free PMC article.
-
Is ftsH the key to plastid longevity in sacoglossan slugs?Genome Biol Evol. 2013;5(12):2540-8. doi: 10.1093/gbe/evt205. Genome Biol Evol. 2013. PMID: 24336424 Free PMC article.
-
Crawling leaves: photosynthesis in sacoglossan sea slugs.J Exp Bot. 2013 Oct;64(13):3999-4009. doi: 10.1093/jxb/ert197. Epub 2013 Jul 11. J Exp Bot. 2013. PMID: 23846876 Review.
-
On Being the Right Size as an Animal with Plastids.Front Plant Sci. 2017 Aug 17;8:1402. doi: 10.3389/fpls.2017.01402. eCollection 2017. Front Plant Sci. 2017. PMID: 28861094 Free PMC article. Review.
Cited by
-
Identification of sequestered chloroplasts in photosynthetic and non-photosynthetic sacoglossan sea slugs (Mollusca, Gastropoda).Front Zool. 2014 Feb 21;11(1):15. doi: 10.1186/1742-9994-11-15. Front Zool. 2014. PMID: 24555467 Free PMC article.
-
Kleptoplast photosynthesis is nutritionally relevant in the sea slug Elysia viridis.Sci Rep. 2017 Aug 10;7(1):7714. doi: 10.1038/s41598-017-08002-0. Sci Rep. 2017. PMID: 28798379 Free PMC article.
-
Chloroplast digestion and the development of functional kleptoplasty in juvenile Elysia timida (Risso, 1818) as compared to short-term and non-chloroplast-retaining sacoglossan slugs.PLoS One. 2017 Oct 11;12(10):e0182910. doi: 10.1371/journal.pone.0182910. eCollection 2017. PLoS One. 2017. PMID: 29020043 Free PMC article.
-
A host organelle integrates stolen chloroplasts for animal photosynthesis.Cell. 2025 Jun 21:S0092-8674(25)00637-3. doi: 10.1016/j.cell.2025.06.003. Online ahead of print. Cell. 2025. PMID: 40570835
-
Mitochondrial Genome Assemblies of Elysia timida and Elysia cornigera and the Response of Mitochondrion-Associated Metabolism during Starvation.Genome Biol Evol. 2017 Jul 1;9(7):1873-1879. doi: 10.1093/gbe/evx129. Genome Biol Evol. 2017. PMID: 28854599 Free PMC article.
References
-
- Venn AA, Loram JE, Douglas AE. 2008. Photosynthetic symbioses in animals. J. Exp. Bot. 59, 1069–1080 (doi:10.1093/jxb/erm328) - DOI - PubMed
-
- Bosch TCG. 2012. What Hydra has to say about the role and origin of symbiotic interactions. Biol. Bull. 223, 78–84 - PubMed
-
- Kovacević G, Franjević D, Jelencić B, Kalafatić M. 2010. Isolation and cultivation of endosymbiotic algae from green Hydra and phylogenetic analysis of 18S rDNA sequences. Folia Biol. 58, 135–143 (doi:10.3409/fb58_1-2.135-143) - DOI - PubMed
-
- Archibald JM. 2009. The puzzle of plastid evolution. Curr. Biol. 19, R81–R88 (doi:10.1016/j.cub.2008.11.067) - DOI - PubMed
-
- Johnson MD. 2011. Acquired phototrophy in ciliates: a review of cellular interactions and structural adaptations. J. Eukaryot. Microbiol. 58, 185–195 (doi:10.1111/j.1550-7408.2011.00545.x) - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

