Evaluating the performance of a rapid antigen test for the detection of influenza virus in clinical specimens from children in Cameroon
- PMID: 24266902
- PMCID: PMC4186458
- DOI: 10.1111/irv.12210
Evaluating the performance of a rapid antigen test for the detection of influenza virus in clinical specimens from children in Cameroon
Abstract
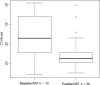
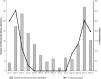
The performance of SD Bioline rapid antigen test (RAT) was evaluated using real-time reverse transcription polymerase chain reaction (rRT-PCR) as gold standard. A total of 718 nasal swabs, including 102 rRT-PCR positive and 616 rRT-PCR negative swabs, were tested. RAT demonstrates a sensitivity of 29·4% with a specificity of 100%. The positivity rate of RAT was highly associated with lower cycle threshold (Ct) values (P < 0·0001). The excellent specificity of the RAT allowed for the rapid identification of influenza cases. However, negative results should be verified by rRT-PCR test because of limitations observed in sensitivity.
Keywords: Cameroon; SD Bioline; influenza; sensitivity; specificity.
© 2013 The Authors. Influenza and Other Respiratory Viruses Published by John Wiley & Sons Ltd.
Figures
Similar articles
-
Clinical validation of a point-of-care multiplexed in vitro immunoassay using monoclonal antibodies (the MSD influenza test) in four hospitals in Vietnam.J Clin Microbiol. 2012 May;50(5):1621-5. doi: 10.1128/JCM.00085-12. Epub 2012 Feb 22. J Clin Microbiol. 2012. PMID: 22357497 Free PMC article.
-
Evaluation of the point-of-care Becton Dickinson Veritor™ Rapid influenza diagnostic test in Kenya, 2013-2014.BMC Infect Dis. 2017 Jan 11;17(1):60. doi: 10.1186/s12879-016-2131-9. BMC Infect Dis. 2017. PMID: 28077093 Free PMC article.
-
Identification of respiratory viruses with a novel point-of-care multianalyte antigen detection test in children with acute respiratory tract infection.J Clin Virol. 2013 Jun;57(2):136-40. doi: 10.1016/j.jcv.2013.02.011. Epub 2013 Mar 11. J Clin Virol. 2013. PMID: 23490399 Free PMC article.
-
Self-collected compared with professional-collected swabbing in the diagnosis of influenza in symptomatic individuals: A meta-analysis and assessment of validity.J Clin Virol. 2019 Sep;118:28-35. doi: 10.1016/j.jcv.2019.07.010. Epub 2019 Aug 2. J Clin Virol. 2019. PMID: 31400670
-
Antigen-based assays for the identification of influenza virus and respiratory syncytial virus: why and how to use them in pediatric practice.Clin Lab Med. 2009 Dec;29(4):649-60. doi: 10.1016/j.cll.2009.07.006. Clin Lab Med. 2009. PMID: 19892226 Review.
Cited by
-
Influence of meteorological parameters in the seasonality of influenza viruses circulating in Northern Cameroon.Influenza Other Respir Viruses. 2019 Mar;13(2):158-165. doi: 10.1111/irv.12612. Epub 2018 Dec 17. Influenza Other Respir Viruses. 2019. PMID: 30220100 Free PMC article.
-
A general approach to identify low-frequency variants within influenza samples collected during routine surveillance.Microb Genom. 2022 Sep;8(9):mgen000867. doi: 10.1099/mgen.0.000867. Microb Genom. 2022. PMID: 36169645 Free PMC article.
-
The epidemiology of seasonal influenza after the 2009 influenza pandemic in Africa: a systematic review.Afr Health Sci. 2020 Dec;20(4):1514-1536. doi: 10.4314/ahs.v20i4.5. Afr Health Sci. 2020. PMID: 34394213 Free PMC article.
References
-
- Simmerman JM, Chittaganpitch M, Erdman D, Sawatwong P, Uyeki TM, Dowell SF. Field performance and new uses of rapid influenza testing in Thailand. Int J Infect Dis. 2007;11:166–171. - PubMed
-
- Rodriguez WJ, Schwartz RH, Thorne MM. Evaluation of diagnostic tests for influenza in a pediatric practice. Pediatr Infect Dis J. 2002;21:193–196. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

