Artemin induced functional recovery and reinnervation after partial nerve injury
- PMID: 24269493
- PMCID: PMC3936608
- DOI: 10.1016/j.pain.2013.11.007
Artemin induced functional recovery and reinnervation after partial nerve injury
Abstract
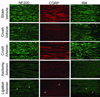
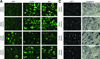
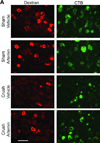
Systemic artemin promotes regeneration of dorsal roots to the spinal cord after crush injury. However, it is unclear whether systemic artemin can also promote peripheral nerve regeneration, and functional recovery after partial lesions distal to the dorsal root ganglion (DRG) remains unknown. In the present investigation, male Sprague Dawley rats received axotomy, ligation, or crush of the L5 spinal nerve or sham surgery. Starting the day of injury, animals received intermittent subcutaneous artemin or vehicle across 2weeks. Sensory thresholds to tactile or thermal stimuli were monitored for 6weeks after injury. Immunohistochemical analyses of the DRG and nerve regeneration were performed at the 6-week time point. Artemin transiently reversed tactile and thermal hypersensitivity after axotomy, ligation, or crush injury. Thermal and tactile hypersensitivity reemerged within 1week of treatment termination. However, artemin-treated rats with nerve crush, but not axotomy or ligation, subsequently showed gradual return of sensory thresholds to preinjury baseline levels by 6weeks after injury. Artemin normalized labeling for NF200, IB4, and CGRP in nerve fibers distal to the crush injury, suggesting persistent normalization of nerve crush-induced neurochemical changes. Sciatic and intradermal administration of dextran or cholera toxin B distal to the crush injury site resulted in labeling of neuronal profiles in the L5 DRG, suggesting regeneration functional restoration of nonmyelinated and myelinated fibers across the injury site into cutaneous tissue. Artemin also diminished ATF3 and caspase 3 expression in the L5 DRG, suggesting persistent neuroprotective actions. A limited period of artemin treatment elicits disease modification by promoting sensory reinnervation of distal territories and restoring preinjury sensory thresholds.
Keywords: Artemin; Hyperalgesia; Peripheral nerve injury; Regeneration; Sensory neurons.
Copyright © 2013 International Association for the Study of Pain. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures





Similar articles
-
Persistent restoration of sensory function by immediate or delayed systemic artemin after dorsal root injury.Nat Neurosci. 2008 Apr;11(4):488-96. doi: 10.1038/nn2069. Epub 2008 Mar 23. Nat Neurosci. 2008. PMID: 18344995 Free PMC article.
-
Neurite outgrowth in normal and injured primary sensory neurons reveals different regulation by nerve growth factor (NGF) and artemin.Mol Cell Neurosci. 2015 Mar;65:125-34. doi: 10.1016/j.mcn.2015.03.004. Epub 2015 Mar 6. Mol Cell Neurosci. 2015. PMID: 25752731
-
Neurotrophin selectivity in organizing topographic regeneration of nociceptive afferents.Exp Neurol. 2015 Sep;271:262-78. doi: 10.1016/j.expneurol.2015.06.007. Epub 2015 Jun 6. Exp Neurol. 2015. PMID: 26054884 Free PMC article.
-
Artemin promotes functional long-distance axonal regeneration to the brainstem after dorsal root crush.Proc Natl Acad Sci U S A. 2015 May 12;112(19):6170-5. doi: 10.1073/pnas.1502057112. Epub 2015 Apr 27. Proc Natl Acad Sci U S A. 2015. PMID: 25918373 Free PMC article.
-
Comparison of neuropathic pain and neuronal apoptosis following nerve root or spinal nerve compression.Eur Spine J. 2009 Dec;18(12):1978-85. doi: 10.1007/s00586-009-1064-z. Epub 2009 Jun 19. Eur Spine J. 2009. PMID: 19543754 Free PMC article.
Cited by
-
Ultrasound imaging of chitosan nerve conduits that bridge sciatic nerve defects in rats.Neural Regen Res. 2014 Jul 15;9(14):1386-8. doi: 10.4103/1673-5374.137592. Neural Regen Res. 2014. PMID: 25221596 Free PMC article. No abstract available.
-
The Neuropeptide Cortistatin Alleviates Neuropathic Pain in Experimental Models of Peripheral Nerve Injury.Pharmaceutics. 2021 Jun 24;13(7):947. doi: 10.3390/pharmaceutics13070947. Pharmaceutics. 2021. PMID: 34202793 Free PMC article.
-
c-Jun gene-modified Schwann cells: upregulating multiple neurotrophic factors and promoting neurite outgrowth.Tissue Eng Part A. 2015 Apr;21(7-8):1409-21. doi: 10.1089/ten.TEA.2014.0416. Tissue Eng Part A. 2015. PMID: 25588149 Free PMC article.
-
Glial cell line-derived neurotrophic factors (GFLs) and small molecules targeting RET receptor for the treatment of pain and Parkinson's disease.Cell Tissue Res. 2020 Oct;382(1):147-160. doi: 10.1007/s00441-020-03227-4. Epub 2020 Jun 17. Cell Tissue Res. 2020. PMID: 32556722 Free PMC article. Review.
-
The role of glial cell line-derived neurotrophic factor family member artemin in neurological disorders and cancers.Cell Prolif. 2020 Jul;53(7):e12860. doi: 10.1111/cpr.12860. Epub 2020 Jun 23. Cell Prolif. 2020. PMID: 32573073 Free PMC article. Review.
References
-
- Allodi I, Udina E, Navarro X. Specificity of peripheral nerve regeneration: interactions at the axon level. Progress in neurobiology. 2012;98(1):16–37. - PubMed
-
- Baloh RH, Tansey MG, Lampe PA, Fahrner TJ, Enomoto H, Simburger KS, Leitner ML, Araki T, Johnson EM, Jr, Milbrandt J. Artemin, a novel member of the GDNF ligand family, supports peripheral and central neurons and signals through the GFRalpha3-RET receptor complex. Neuron. 1998;21(6):1291–1302. - PubMed
-
- Bennett DL, Boucher TJ, Armanini MP, Poulsen KT, Michael GJ, Priestley JV, Phillips HS, McMahon SB, Shelton DL. The glial cell line-derived neurotrophic factor family receptor components are differentially regulated within sensory neurons after nerve injury. J Neurosci. 2000;20(1):427–437. - PMC - PubMed
-
- Bennett DL, Boucher TJ, Michael GJ, Popat RJ, Malcangio M, Averill SA, Poulsen KT, Priestley JV, Shelton DL, McMahon SB. Artemin has potent neurotrophic actions on injured C-fibres. J Peripher Nerv Syst. 2006;11(4):330–345. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

