Bacterial cell-envelope glycoconjugates
- PMID: 24274370
- PMCID: PMC4396862
- DOI: 10.1016/B978-0-12-408093-5.00006-X
Bacterial cell-envelope glycoconjugates
Abstract
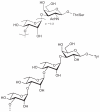
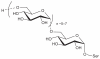
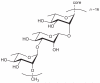
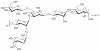
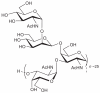
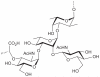
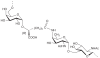
Prokaryotic glycosylation fulfills an important role in maintaining and protecting the structural integrity and function of the bacterial cell wall, as well as serving as a flexible adaption mechanism to evade environmental and host-induced pressure. The scope of bacterial and archaeal protein glycosylation has considerably expanded over the past decade(s), with numerous examples covering the glycosylation of flagella, pili, glycosylated enzymes, as well as surface-layer proteins. This article addresses structure, analysis, function, genetic basis, biosynthesis, and biomedical and biotechnological applications of cell-envelope glycoconjugates, S-layer glycoprotein glycans, and "nonclassical" secondary-cell wall polysaccharides. The latter group of polymers mediates the important attachment and regular orientation of the S-layer to the cell wall. The structures of these glycopolymers reveal an enormous diversity, resembling the structural variability of bacterial lipopolysaccharides and capsular polysaccharides. While most examples are presented for Gram-positive bacteria, the S-layer glycan of the Gram-negative pathogen Tannerella forsythia is also discussed. In addition, archaeal S-layer glycoproteins are briefly summarized.
Keywords: Archaea; Bacteria; Glycoprotein; Secondary cell-wall polymers; Surface layer.
© 2013 Elsevier Inc. All rights reserved.
Figures

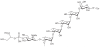
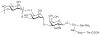





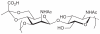
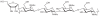
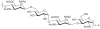

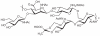
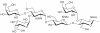
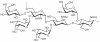
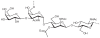


References
-
- Kobata A. The carbohydrates of glycoproteins. In: Ginsburg V, Robbins PW, editors. Biology of Carbohydrates. Vol. 2. John Wiley & Sons; New York: 1984. pp. 87–161.
-
- Montreuil J. Spatial conformation of glycans and glycoproteins. Biol. Cell. 1984;51:115–131. - PubMed
-
- Kornfeld R, Kornfeld S. Assembly of asparagine-linked oligosaccharides. Annu. Rev. Biochem. 1985;54:631–664. - PubMed
-
- Varki A, Cummings RD, Esko JD, Freeze HH, Stanley P, Bertozzi CR, Hart GW, Etzler ME, editors. Essentials of Glycobiology. 2nd ed. Cold Spring Harbor Laboratory Press; Cold Spring Harbor, NY: 2009. p. 784. - PubMed
-
- Messner P, Sleytr UB. Bacterial surface layer glycoproteins. Glycobiology. 1991;1:545–551. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

