Studies on the expression of sesquiterpene synthases using promoter-β-glucuronidase fusions in transgenic Artemisia annua L
- PMID: 24278301
- PMCID: PMC3838408
- DOI: 10.1371/journal.pone.0080643
Studies on the expression of sesquiterpene synthases using promoter-β-glucuronidase fusions in transgenic Artemisia annua L
Abstract
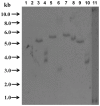
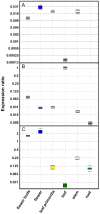
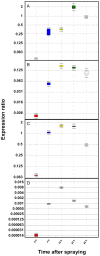
In order to better understand the influence of sesquiterpene synthases on artemisinin yield in Artemisia annua, the expression of some sesquiterpene synthases has been studied using transgenic plants expressing promoter-GUS fusions. The cloned promoter sequences were 923, 1182 and 1510 bp for β-caryophyllene (CPS), epi-cedrol (ECS) and β-farnesene (FS) synthase, respectively. Prediction of cis-acting regulatory elements showed that the promoters are involved in complex regulation of expression. Transgenic A. annua plants carrying promoter-GUS fusions were studied to elucidate the expression pattern of the three sesquiterpene synthases and compared to the previously studied promoter of amorpha-4,11-diene synthase (ADS), a key enzyme of artemisinin biosynthesis. The CPS and ECS promoters were active in T-shaped trichomes of leaves and stems, basal bracts of flower buds and also in some florets cells but not in glandular secretory trichome while FS promoter activity was only observed in leaf cells and trichomes of transgenic shoots. ADS, CPS, ECS and FS transcripts were induced by wounding in a time depended manner. The four sesquiterpene synthases may be involved in responsiveness of A. annua to herbivory. Methyl jasmonate treatment triggered activation of the promoters of all four sesquiterpene synthases in a time depended manner. Southern blot result showed that the GUS gene was inserted into genomic DNA of transgenic lines as a single copy or two copies. The relative amounts of CPS and ECS as well as germacrene A synthase (GAS) transcripts are much lower than that of ADS transcript. Consequently, down-regulation of the expression of the CPS, ECS or GAS gene may not improve artemsinin yield. However, blocking the expression of FS may have effects on artemisinin production.
Conflict of interest statement
Figures









Similar articles
-
Studies on the expression of linalool synthase using a promoter-β-glucuronidase fusion in transgenic Artemisia annua.J Plant Physiol. 2014 Jan 15;171(2):85-96. doi: 10.1016/j.jplph.2013.09.019. Epub 2013 Nov 16. J Plant Physiol. 2014. PMID: 24331423
-
Trichome-specific expression of the amorpha-4,11-diene 12-hydroxylase (cyp71av1) gene, encoding a key enzyme of artemisinin biosynthesis in Artemisia annua, as reported by a promoter-GUS fusion.Plant Mol Biol. 2013 Jan;81(1-2):119-38. doi: 10.1007/s11103-012-9986-y. Epub 2012 Nov 19. Plant Mol Biol. 2013. PMID: 23161198
-
T-shaped trichome-specific expression of monoterpene synthase ADH2 using promoter-β-GUS fusion in transgenic Artemisia annua L.Biotechnol Appl Biochem. 2016 Nov;63(6):834-840. doi: 10.1002/bab.1440. Epub 2015 Nov 5. Biotechnol Appl Biochem. 2016. PMID: 26340706
-
[Advances in sesquiterpene synthases cyclases of Artemisia annua].Sheng Wu Gong Cheng Xue Bao. 2007 Nov;23(6):976-81. doi: 10.1016/s1872-2075(07)60059-4. Sheng Wu Gong Cheng Xue Bao. 2007. PMID: 18257222 Review. Chinese.
-
[Advances in molecular regulation of artemisinin biosynthesis].Sheng Wu Gong Cheng Xue Bao. 2003 Nov;19(6):646-50. Sheng Wu Gong Cheng Xue Bao. 2003. PMID: 15971573 Review. Chinese.
Cited by
-
Aphid alarm pheromone mimicry in transgenic Chrysanthemum morifolium: insights into the potential of (E)-β-farnesene for aphid resistance.Front Plant Sci. 2024 Apr 22;15:1373669. doi: 10.3389/fpls.2024.1373669. eCollection 2024. Front Plant Sci. 2024. PMID: 38711605 Free PMC article.
-
A Dual Repeat Cis-Element Determines Expression of GERANYL DIPHOSPHATE SYNTHASE for Monoterpene Production in Phalaenopsis Orchids.Front Plant Sci. 2018 Jun 5;9:765. doi: 10.3389/fpls.2018.00765. eCollection 2018. Front Plant Sci. 2018. PMID: 29922327 Free PMC article.
-
AaPDR3, a PDR Transporter 3, Is Involved in Sesquiterpene β-Caryophyllene Transport in Artemisia annua.Front Plant Sci. 2017 May 8;8:723. doi: 10.3389/fpls.2017.00723. eCollection 2017. Front Plant Sci. 2017. PMID: 28533790 Free PMC article.
-
Multisite evaluation of phenotypic plasticity for specialized metabolites, some involved in carrot quality and disease resistance.PLoS One. 2021 Apr 2;16(4):e0249613. doi: 10.1371/journal.pone.0249613. eCollection 2021. PLoS One. 2021. PMID: 33798246 Free PMC article.
-
Enhancing epi-cedrol production in Escherichia coli by fusion expression of farnesyl pyrophosphate synthase and epi-cedrol synthase.Eng Life Sci. 2019 Jul 25;19(9):606-616. doi: 10.1002/elsc.201900103. eCollection 2019 Sep. Eng Life Sci. 2019. PMID: 32625036 Free PMC article.
References
-
- Cai Y, Jia J-W, Crock J, Lin Z-X, Chen X-Y, et al. (2002) A cDNA clone for β-caryophyllene synthase from Artemisia annua . Phytochemistry 61: 523–529. - PubMed
-
- Cheng A-X, Xiang C-Y, Li J-X, Yang C-Q, Hu W-L, et al. (2007) The rice (E)-β-caryophyllene synthase (OsTPS3) accounts for the major inducible volatile sesquiterpenes. Phytochemistry 68: 1632–1641. - PubMed
-
- Goel D, Goel R, Singh V, Ali M, Mallavarapu GR, et al. (2007) Composition of the essential oil from the root of Artemisia annua . J Nat Med 61: 458–461.
-
- Bilia AR, Flamini G, Morgenni F, Isacchi B, Vincieri FF (2008) GC MS analysis of the volatile constituents of essential oil and aromatic waters of Artemisia annua L. at different developmental stages. Nat Prod Commun 3: 2075–2078.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

