Glycosylation of the Escherichia coli TibA self-associating autotransporter influences the conformation and the functionality of the protein
- PMID: 24278316
- PMCID: PMC3835316
- DOI: 10.1371/journal.pone.0080739
Glycosylation of the Escherichia coli TibA self-associating autotransporter influences the conformation and the functionality of the protein
Abstract
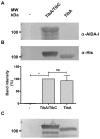
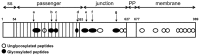
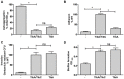
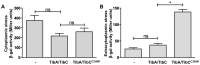
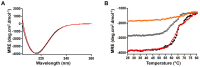
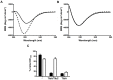
The self-associating autotransporters (SAATs) are multifunctional secreted proteins of Escherichia coli, comprising the AIDA-I, TibA and Ag43 proteins. One of their characteristics is that they can be glycosylated. Glycosylation of AIDA-I and Ag43 have been investigated, but not that of TibA. It is still not clear whether glycosylation of the SAATs affect their structure or their functionality. Therefore, we have looked at the effects of glycosylation on the TibA adhesin/invasin. TibA is glycosylated by TibC, a specific glycosyltransferase, and the two genes are encoded in an operon. In this study, we have found that the glycosylation of TibA is not limited to the extracellular functional domain, as previously observed with AIDA-I and Ag43. We have determined that unglycosylated TibA is not able to promote the adhesion of bacteria on cultured epithelial cell, even though it is still able to promote invasion, biofilm formation and autoaggregation of bacteria. We have purified the glycosylated and unglycosylated forms of TibA, and determined that TibA is less stable when not glycosylated. We finally observed that glycosylation affects the oligomerisation of TibA and that unglycosylated TibA is locked in a conformation that is not suited for adhesion. Our results suggest that the effect of glycosylation on the functionality of TibA is indirect.
Conflict of interest statement
Figures

Similar articles
-
O-linked glycosylation ensures the normal conformation of the autotransporter adhesin involved in diffuse adherence.J Bacteriol. 2007 Dec;189(24):8880-9. doi: 10.1128/JB.00969-07. Epub 2007 Oct 19. J Bacteriol. 2007. PMID: 17951390 Free PMC article.
-
Conformation change in a self-recognizing autotransporter modulates bacterial cell-cell interaction.J Biol Chem. 2010 Apr 2;285(14):10616-26. doi: 10.1074/jbc.M109.069070. Epub 2010 Feb 1. J Biol Chem. 2010. PMID: 20123991 Free PMC article.
-
Structure-function analysis of the TibA self-associating autotransporter reveals a modular organization.Infect Immun. 2011 May;79(5):1826-32. doi: 10.1128/IAI.01129-10. Epub 2011 Feb 22. Infect Immun. 2011. PMID: 21343356 Free PMC article.
-
Self-associating autotransporters, SAATs: functional and structural similarities.Int J Med Microbiol. 2006 Aug;296(4-5):187-95. doi: 10.1016/j.ijmm.2005.10.002. Int J Med Microbiol. 2006. PMID: 16600681 Review.
-
Autotransporter Adhesins in Escherichia coli Pathogenesis.Proteomics. 2017 Dec;17(23-24). doi: 10.1002/pmic.201600431. Epub 2017 Oct 12. Proteomics. 2017. PMID: 28665015 Review.
Cited by
-
Opportunities and Challenges of Bacterial Glycosylation for the Development of Novel Antibacterial Strategies.Front Microbiol. 2021 Sep 24;12:745702. doi: 10.3389/fmicb.2021.745702. eCollection 2021. Front Microbiol. 2021. PMID: 34630370 Free PMC article. Review.
-
Processivity in Bacterial Glycosyltransferases.ACS Chem Biol. 2020 Jan 17;15(1):3-16. doi: 10.1021/acschembio.9b00619. Epub 2019 Dec 11. ACS Chem Biol. 2020. PMID: 31750644 Free PMC article. Review.
-
Two-Partner Secretion: Combining Efficiency and Simplicity in the Secretion of Large Proteins for Bacteria-Host and Bacteria-Bacteria Interactions.Front Cell Infect Microbiol. 2017 May 9;7:148. doi: 10.3389/fcimb.2017.00148. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28536673 Free PMC article. Review.
-
Global landscape of cell envelope protein complexes in Escherichia coli.Nat Biotechnol. 2018 Jan;36(1):103-112. doi: 10.1038/nbt.4024. Epub 2017 Nov 27. Nat Biotechnol. 2018. PMID: 29176613 Free PMC article.
-
The N-linking glycosylation system from Actinobacillus pleuropneumoniae is required for adhesion and has potential use in glycoengineering.Open Biol. 2017 Jan;7(1):160212. doi: 10.1098/rsob.160212. Open Biol. 2017. PMID: 28077594 Free PMC article.
References
-
- Nothaft H, Szymanski CM (2010) Protein glycosylation in bacteria: sweeter than ever. Nat Rev Microbiol 8: 765–778. - PubMed
-
- Schmidt MA, Riley LW, Benz I (2003) Sweet new world: glycoproteins in bacterial pathogens. Trends Microbiol 11: 554–561. - PubMed
-
- Szymanski CM, Wren BW (2005) Protein glycosylation in bacterial mucosal pathogens. Nat Rev Microbiol 3: 225–237. - PubMed
-
- Szymanski CM, Logan SM, Linton D, Wren BW (2003) Campylobacter–a tale of two protein glycosylation systems. Trends Microbiol 11: 233–238. - PubMed
-
- Scott NE, Parker BL, Connolly AM, Paulech J, Edwards AV, et al. (2011) Simultaneous glycan-peptide characterization using hydrophilic interaction chromatography and parallel fragmentation by CID, higher energy collisional dissociation, and electron transfer dissociation MS applied to the N-linked glycoproteome of Campylobacter jejuni. Mol Cell Proteomics 10: M000031–MCP000201. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

