Feasibility of the use of combinatorial chemokine arrays to study blood and CSF in multiple sclerosis
- PMID: 24278364
- PMCID: PMC3835751
- DOI: 10.1371/journal.pone.0081007
Feasibility of the use of combinatorial chemokine arrays to study blood and CSF in multiple sclerosis
Abstract
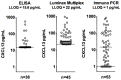
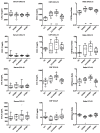
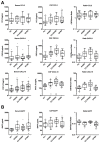
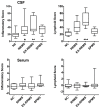
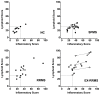
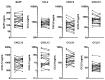
Meningeal inflammation, including the presence of semi-organized tertiary lymphoid tissue, has been associated with cortical pathology at autopsy in secondary progressive multiple sclerosis (SPMS). Accessible and robust biochemical markers of cortical inflammation for use in SPMS clinical trials are needed. Increased levels of chemokines in the cerebrospinal fluid (CSF) can report on inflammatory processes occurring in the cerebral cortex of MS patients. A multiplexed chemokine array that included BAFF, a high sensitivity CXCL13 assay and composite chemokine scores were developed to explore differences in lymphoid (CXCL12, CXCL13, CCL19 and CCL21) and inflammatory (CCL2, CXCL9, CXCL10 and CXCL11) chemokines in a small pilot study. Paired CSF and serum samples were obtained from healthy controls (n=12), relapsing-remitting MS (RRMS) (n=21) and SPMS (N=12). A subset of the RRMS patients (n = 9) was assessed upon disease exacerbation and 1 month later following iv methylprednisone. SPMS patients were sampled twice to ascertain stability. Both lymphoid and inflammatory chemokines were elevated in RRMS and SPMS with the highest levels found in the active RRMS group. Inflammatory and lymphoid chemokine signatures were defined and generally correlated with each other. This small exploratory clinical study shows the feasibility of measuring complex and potentially more robust chemokine signatures in the CSF of MS patients during clinical trials. No differences were found between stable RRMS and SPMS. Future trials with larger patient cohorts with this chemokine array are needed to further characterize the differences, or the lack thereof, between stable RRMS and SPMS.
Conflict of interest statement
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

