The olfactory bulb in newborn piglet is a reservoir of neural stem and progenitor cells
- PMID: 24278384
- PMCID: PMC3836747
- DOI: 10.1371/journal.pone.0081105
The olfactory bulb in newborn piglet is a reservoir of neural stem and progenitor cells
Abstract
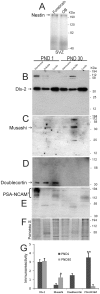
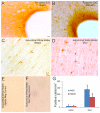
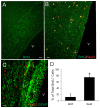
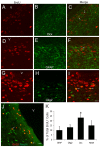
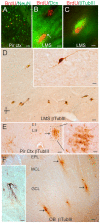
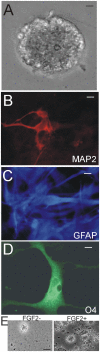
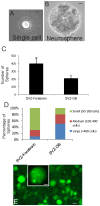
The olfactory bulb (OB) periventricular zone is an extension of the forebrain subventricular zone (SVZ) and thus is a source of neuroprogenitor cells and neural stem cells. While considerable information is available on the SVZ-OB neural stem cell (NSC)/neuroprogenitor cell (NPC) niche in rodents, less work has been done on this system in large animals. The newborn piglet is used as a preclinical translational model of neonatal hypoxic-ischemic brain damage, but information about the endogenous sources of NSCs/NPCs in piglet is needed to implement endogenous or autologous cell-based therapies in this model. We characterized NSC/NPC niches in piglet forebrain and OB-SVZ using western blotting, histological, and cell culture methods. Immunoblotting revealed nestin, a NSC/NPC marker, in forebrain-SVZ and OB-SVZ in newborn piglet. Several progenitor or newborn neuron markers, including Dlx2, musashi, doublecortin, and polysialated neural cell adhesion molecule were also detected in OB-SVZ by immunoblotting. Immunohistochemistry confirmed the presence of nestin, musashi, and doublecortin in forebrain-SVZ and OB-SVZ. Bromodeoxyuridine (BrdU) labeling showed that the forebrain-SVZ and OB-SVZ accumulate newly replicated cells. BrdU-positive cells were immunolabeled for astroglial, oligodendroglial, and neuronal markers. A lateral migratory pathway for newly born neuron migration to primary olfactory cortex was revealed by BrdU labeling and co-labeling for doublecortin and class III β tubulin. Isolated and cultured forebrain-SVZ and OB-SVZ cells from newborn piglet had the capacity to generate numerous neurospheres. Single cell clonal analysis of neurospheres revealed the capacity for self-renewal and multipotency. Neurosphere-derived cells differentiated into neurons, astrocytes, and oligodendrocytes and were amenable to permanent genetic tagging with lentivirus encoding green fluorescent protein. We conclude that the piglet OB-SVZ is a reservoir of NSCs and NPCs suitable to use in autologous cell therapy in preclinical models of neonatal/pediatric brain injury.
Conflict of interest statement
Figures


Similar articles
-
Olfactory bulb core is a rich source of neural progenitor and stem cells in adult rodent and human.J Comp Neurol. 2003 May 12;459(4):368-91. doi: 10.1002/cne.10664. J Comp Neurol. 2003. PMID: 12687705
-
Neonatal SVZ EGFP-labeled cells produce neurons in the olfactory bulb and astrocytes in the cerebral cortex by in-vivo electroporation.Neuroreport. 2013 May 8;24(7):381-7. doi: 10.1097/WNR.0b013e328360f7ef. Neuroreport. 2013. PMID: 23568218
-
Ketamine interferes with the proliferation and differentiation of neural stem cells in the subventricular zone of neonatal rats.Cell Physiol Biochem. 2015;35(1):315-25. doi: 10.1159/000369698. Epub 2015 Jan 10. Cell Physiol Biochem. 2015. PMID: 25591773
-
The Adult Ventricular-Subventricular Zone (V-SVZ) and Olfactory Bulb (OB) Neurogenesis.Cold Spring Harb Perspect Biol. 2016 May 2;8(5):a018820. doi: 10.1101/cshperspect.a018820. Cold Spring Harb Perspect Biol. 2016. PMID: 27048191 Free PMC article. Review.
-
The heterogeneity of adult neural stem cells and the emerging complexity of their niche.Cold Spring Harb Symp Quant Biol. 2008;73:357-65. doi: 10.1101/sqb.2008.73.019. Epub 2008 Nov 6. Cold Spring Harb Symp Quant Biol. 2008. PMID: 19022766 Review.
Cited by
-
DNA Damage Response and Repair, DNA Methylation, and Cell Death in Human Neurons and Experimental Animal Neurons Are Different.J Neuropathol Exp Neurol. 2018 Jul 1;77(7):636-655. doi: 10.1093/jnen/nly040. J Neuropathol Exp Neurol. 2018. PMID: 29788379 Free PMC article.
-
Single-Cell Sequencing of Glioblastoma Reveals Central Nervous System Susceptibility to SARS-CoV-2.Front Oncol. 2020 Nov 16;10:566599. doi: 10.3389/fonc.2020.566599. eCollection 2020. Front Oncol. 2020. PMID: 33312949 Free PMC article.
-
Proteasome localization and activity in pig brain and in vivo small molecule screening for activators.Front Cell Neurosci. 2024 Feb 26;18:1353542. doi: 10.3389/fncel.2024.1353542. eCollection 2024. Front Cell Neurosci. 2024. PMID: 38469354 Free PMC article.
-
Hypothermic Protection in Neocortex Is Topographic and Laminar, Seizure Unmitigating, and Partially Rescues Neurons Depleted of RNA Splicing Protein Rbfox3/NeuN in Neonatal Hypoxic-Ischemic Male Piglets.Cells. 2023 Oct 15;12(20):2454. doi: 10.3390/cells12202454. Cells. 2023. PMID: 37887298 Free PMC article.
-
The Pig Olfactory Brain: A Primer.Chem Senses. 2016 Jun;41(5):415-25. doi: 10.1093/chemse/bjw016. Epub 2016 Mar 2. Chem Senses. 2016. PMID: 26936231 Free PMC article.
References
-
- Ikeda T, Iwai M, Hayashi T, Nagano I, Shogi M et al. (2005) Limited differentiation to neurons and astroglia from neural stem cells in the cortex and striatum after ischemia/hypoxia in the neonatal rat brain. Am J Obstet Gynecol 193: 849-856. doi:10.1016/j.ajog.2005.01.029. PubMed: 16150285. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

