Use of induced pluripotent stem cells to recapitulate pulmonary alveolar proteinosis pathogenesis
- PMID: 24279752
- PMCID: PMC3983889
- DOI: 10.1164/rccm.201306-1039OC
Use of induced pluripotent stem cells to recapitulate pulmonary alveolar proteinosis pathogenesis
Abstract
Rationale: In patients with pulmonary alveolar proteinosis (PAP) syndrome, disruption of granulocyte/macrophage colony-stimulating factor (GM-CSF) signaling is associated with pathogenic surfactant accumulation from impaired clearance in alveolar macrophages.
Objectives: The aim of this study was to overcome these barriers by using monocyte-derived induced pluripotent stem (iPS) cells to recapitulate disease-specific and normal macrophages.
Methods: We created iPS cells from two children with hereditary PAP (hPAP) caused by recessive CSF2RA(R217X) mutations and three normal people, differentiated them into macrophages (hPAP-iPS-Mφs and NL-iPS-Mφs, respectively), and evaluated macrophage functions with and without gene-correction to restore GM-CSF signaling in hPAP-iPS-Mφs.
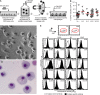
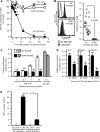
Measurements and main results: Both hPAP and normal iPS cells had human embryonic stem cell-like morphology, expressed pluripotency markers, formed teratomas in vivo, had a normal karyotype, retained and expressed mutant or normal CSF2RA genes, respectively, and could be differentiated into macrophages with the typical morphology and phenotypic markers. Compared with normal, hPAP-iPS-Mφs had impaired GM-CSF receptor signaling and reduced GM-CSF-dependent gene expression, GM-CSF- but not M-CSF-dependent cell proliferation, surfactant clearance, and proinflammatory cytokine secretion. Restoration of GM-CSF receptor signaling corrected the surfactant clearance abnormality in hPAP-iPS-Mφs.
Conclusions: We used patient-specific iPS cells to accurately reproduce the molecular and cellular defects of alveolar macrophages that drive the pathogenesis of PAP in more than 90% of patients. These results demonstrate the critical role of GM-CSF signaling in surfactant homeostasis and PAP pathogenesis in humans and have therapeutic implications for hPAP.
Figures


Comment in
-
Modeling pulmonary alveolar proteinosis with induced pluripotent stem cells.Am J Respir Crit Care Med. 2014 Jan 15;189(2):124-6. doi: 10.1164/rccm.201312-2122ED. Am J Respir Crit Care Med. 2014. PMID: 24428646 No abstract available.
Similar articles
-
Gene correction of human induced pluripotent stem cells repairs the cellular phenotype in pulmonary alveolar proteinosis.Am J Respir Crit Care Med. 2014 Jan 15;189(2):167-82. doi: 10.1164/rccm.201306-1012OC. Am J Respir Crit Care Med. 2014. PMID: 24279725
-
A murine model of hereditary pulmonary alveolar proteinosis caused by homozygous Csf2ra gene disruption.Am J Physiol Lung Cell Mol Physiol. 2022 Mar 1;322(3):L438-L448. doi: 10.1152/ajplung.00175.2021. Epub 2022 Jan 19. Am J Physiol Lung Cell Mol Physiol. 2022. PMID: 35043685 Free PMC article.
-
Function and Safety of Lentivirus-Mediated Gene Transfer for CSF2RA-Deficiency.Hum Gene Ther Methods. 2017 Dec;28(6):318-329. doi: 10.1089/hgtb.2017.092. Epub 2017 Aug 30. Hum Gene Ther Methods. 2017. PMID: 28854814 Free PMC article.
-
The molecular basis of pulmonary alveolar proteinosis.Clin Immunol. 2010 May;135(2):223-35. doi: 10.1016/j.clim.2010.02.017. Epub 2010 Mar 25. Clin Immunol. 2010. PMID: 20338813 Free PMC article. Review.
-
Immune dysregulation in the pathogenesis of pulmonary alveolar proteinosis.Curr Allergy Asthma Rep. 2010 Sep;10(5):320-5. doi: 10.1007/s11882-010-0134-y. Curr Allergy Asthma Rep. 2010. PMID: 20623372 Review.
Cited by
-
Murine iPSC-Derived Macrophages as a Tool for Disease Modeling of Hereditary Pulmonary Alveolar Proteinosis due to Csf2rb Deficiency.Stem Cell Reports. 2016 Aug 9;7(2):292-305. doi: 10.1016/j.stemcr.2016.06.011. Epub 2016 Jul 21. Stem Cell Reports. 2016. PMID: 27453007 Free PMC article.
-
Lung regeneration: mechanisms, applications and emerging stem cell populations.Nat Med. 2014 Aug;20(8):822-32. doi: 10.1038/nm.3642. Nat Med. 2014. PMID: 25100528 Free PMC article. Review.
-
Ex vivo Generation of Genetically Modified Macrophages from Human Induced Pluripotent Stem Cells.Transfus Med Hemother. 2017 Jun;44(3):135-142. doi: 10.1159/000477129. Epub 2017 May 16. Transfus Med Hemother. 2017. PMID: 28626364 Free PMC article.
-
Diseases of pulmonary surfactant homeostasis.Annu Rev Pathol. 2015;10:371-93. doi: 10.1146/annurev-pathol-012513-104644. Annu Rev Pathol. 2015. PMID: 25621661 Free PMC article. Review.
-
Forthcoming complications in recovered COVID-19 patients with COPD and asthma; possible therapeutic opportunities.Cell Commun Signal. 2022 Nov 1;20(1):173. doi: 10.1186/s12964-022-00982-5. Cell Commun Signal. 2022. PMID: 36320055 Free PMC article. Review.
References
-
- Suzuki T, Maranda B, Sakagami T, Catellier P, Couture CY, Carey BC, Chalk C, Trapnell BC. Hereditary pulmonary alveolar proteinosis caused by recessive CSF2RB mutations. Eur Respir J. 2011;37:201–204. - PubMed
-
- Tanaka T, Motoi N, Tsuchihashi Y, Tazawa R, Kaneko C, Nei T, Yamamoto T, Hayashi T, Tagawa T, Nagayasu T, et al. Adult-onset hereditary pulmonary alveolar proteinosis caused by a single-base deletion in CSF2RB. J Med Genet. 2011;48:205–209. - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

