Aralia cordata inhibits triacylglycerol biosynthesis in HepG2 cells
- PMID: 24283275
- PMCID: PMC3868379
- DOI: 10.1089/jmf.2012.2636
Aralia cordata inhibits triacylglycerol biosynthesis in HepG2 cells
Abstract
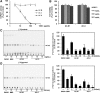
Glycerol-3-phosphate acyltransferase (GPAT) catalyzes the first committed step in triacylglycerol (TAG) and phospholipid biosynthesis, and has been considered as one of the drug targets for treating hepatic steatosis, insulin resistance, and other metabolic disorders. The aim of this study was to investigate the GPAT inhibitors from natural products and to evaluate their effects. The methanol extract of Aralia cordata roots showed a strong inhibitory effect on the human GPAT1 activity. A further bioactivity-guided approach led to the isolation of ent-pimara-8(14),15-dien-19-oic acid, (PA), one of the major compounds of A. cordata, which suppressed the GPAT1 activity with IC50 value of 60.5 μM. PA markedly reduced de novo lysophosphatidic acid synthesis through inhibition of GPAT activity and therefore significantly decreased synthesis of TAG in the HepG2 cells. These results suggest that PA as well as A. cordata root extract could be beneficial in controlling lipid metabolism.
Figures



Similar articles
-
Cholinesterase and BACE1 inhibitory diterpenoids from Aralia cordata.Arch Pharm Res. 2009 Oct;32(10):1399-408. doi: 10.1007/s12272-009-2009-0. Epub 2009 Nov 8. Arch Pharm Res. 2009. PMID: 19898803
-
Cytotoxic and DNA topoisomerases I and II inhibitory constituents from the roots of Aralia cordata.Arch Pharm Res. 2007 Nov;30(11):1404-9. doi: 10.1007/BF02977364. Arch Pharm Res. 2007. PMID: 18087808
-
Inhibitory activity of Aralia continentalis roots on protein tyrosine phosphatase 1B and rat lens aldose reductase.Arch Pharm Res. 2012 Oct;35(10):1771-7. doi: 10.1007/s12272-012-1009-7. Epub 2012 Nov 9. Arch Pharm Res. 2012. PMID: 23139128
-
Regulation of Triglyceride Metabolism. II. Function of mitochondrial GPAT1 in the regulation of triacylglycerol biosynthesis and insulin action.Am J Physiol Gastrointest Liver Physiol. 2007 May;292(5):G1195-9. doi: 10.1152/ajpgi.00553.2006. Epub 2006 Dec 7. Am J Physiol Gastrointest Liver Physiol. 2007. PMID: 17158253 Free PMC article. Review.
-
Glycerol-3-phosphate acyltransferases: rate limiting enzymes of triacylglycerol biosynthesis.Biochim Biophys Acta. 2009 Jun;1791(6):501-6. doi: 10.1016/j.bbalip.2008.10.010. Epub 2008 Nov 7. Biochim Biophys Acta. 2009. PMID: 19038363 Free PMC article. Review.
Cited by
-
Aralia armata (Wall.) Seem Improves Intimal Hyperplasia after Vascular Injury by Downregulating the Wnt3α/Dvl-1/β-Catenin Pathway.Biomed Res Int. 2021 Jul 12;2021:6682525. doi: 10.1155/2021/6682525. eCollection 2021. Biomed Res Int. 2021. PMID: 34337044 Free PMC article.
-
Aralia cordata Thunb. as a Source of Bioactive Compounds: Phytochemical Composition and Antioxidant Activity.Plants (Basel). 2022 Jun 28;11(13):1704. doi: 10.3390/plants11131704. Plants (Basel). 2022. PMID: 35807656 Free PMC article.
References
-
- Tziomalos K, Athyros VG, Karagiannis A, Kolovou GD, Mikhailidis DP: Triglycerides and vascular risk: insights from epidemiological data and interventional studies. Curr Drug Targets 2009;10:320–327 - PubMed
-
- Hammond LE, Neschen S, Romanelli AJ, et al. .: Mitochondrial glycerol-3-phosphate acyltransferase-1 is essential in liver for the metabolism of excess acyl-CoAs. J Biol Chem 2005;280:25629–25636 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

