Isolation and characterization of collagen and antioxidant collagen peptides from scales of croceine croaker (Pseudosciaena crocea)
- PMID: 24284428
- PMCID: PMC3853751
- DOI: 10.3390/md11114641
Isolation and characterization of collagen and antioxidant collagen peptides from scales of croceine croaker (Pseudosciaena crocea)
Abstract
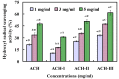
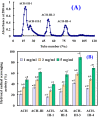
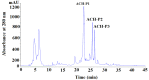
Acid soluble collagen (ASC) from scales of croceine croaker (ASC-C) was successfully isolated with the yield of 0.37% ± 0.08% (dry weight basis), and characterized as type I collagen on the basis of amino acid analysis and electrophoretic pattern. The antioxidant hydrolysate of ASC-C (ACH) was prepared through a two-stage in vitro digestion (4-h trypsin followed by 4-h pepsin), and three antioxidant peptides (ACH-P1, ACH-P2, and ACH-P3) were further isolated from ACH using ultrafiltration, gel chromatography, and RP-HPLC, and their amino acid sequences were identified as GFRGTIGLVG (ACH-P1), GPAGPAG (ACH-P2), and GFPSG (ACH-P3). ACH-P1, ACH-P2, and ACH-P3 showed good scavenging activities on hydroxyl radical (IC50 0.293, 0.240, and 0.107 mg/mL, respectively), DPPH radical (IC₅₀ 1.271, 0.675, and 0.283 mg/mL, respectively), superoxide radical (IC₅₀ 0.463, 0.099, and 0.151 mg/mL, respectively), and ABTS radical (IC₅₀ 0.421, 0.309, and 0.210 mg/mL, respectively). ACH-P3 was also effectively against lipid peroxidation in the model system. The antioxidant activities of three collagen peptides were due to the presence of hydrophobic amino acid residues within the peptide sequences. The collagen peptides might be used as antioxidant for the therapy of diseases associated with oxidative stress, or reducing oxidative changes during storage.
Figures



References
-
- Sampath Kumar N.S., Nazeer R.A., Jaiganesh R. Purification and identification of antioxidant peptides from the skin protein hydrolysate of two marine fishes, horse mackerel (Magalaspis cordyla) and croaker (Otolithes ruber) Amino Acids. 2012;42:1641–1649. doi: 10.1007/s00726-011-0858-6. - DOI - PubMed
-
- Lee W.S., Jeon J.K., Byun H.G. Characterization of a novel antioxidative peptide from the sand eel Hypoptychus dybowskii. Proc. Biochem. 2011;46:1207–1211. doi: 10.1016/j.procbio.2011.02.001. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

