The cataract-associated V41M mutant of human γS-crystallin shows specific structural changes that directly enhance local surface hydrophobicity
- PMID: 24287181
- PMCID: PMC3927989
- DOI: 10.1016/j.bbrc.2013.11.073
The cataract-associated V41M mutant of human γS-crystallin shows specific structural changes that directly enhance local surface hydrophobicity
Abstract
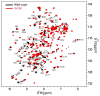
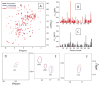
The major crystallins expressed in the human lens are γS-, γC- and γD-crystallins. Several mutations in γS-crystallin are associated with hereditary cataracts, one of which involves the substitution of a highly conserved Valine at position 41 to Methionine. According to a recent report, the mutant protein, V41M, shows lower stability and increased surface hydrophobicity compared to the wild-type, and a propensity for self-aggregation. Here we address the structural differences between the two proteins, with residue-level specificity using NMR spectroscopy. Based on the structural model of the mutant protein, our results clearly show that the mutation creates a major local perturbation almost at the junction of the first and second "Greek-key" motifs in the N-terminal domain. A larger section of the second motif (residues 44-86) appears to be mainly affected. Based on the sizeable chemical shift of the imino proton of the indole side-chain of Trp46 in V41M, we suggest that the sulphur atom of Met41 is involved in an S-π interaction with Trp46. This interaction would bring the last β-strand of the first "Greek-key" motif closer to the first β-strand of the second motif. This appears to lead to a domino effect, towards both the N- and C-terminal ends, even as it decays off substantially beyond the domain interface. During this process discreet hydrophobic surface patches are created, as revealed by ANS-binding. Such changes would not affect the secondary structure or cause a major change in the tertiary structure, but can lead to self-aggregation or aberrant binding interactions of the mutant protein in vivo, and lead to lens opacity or cataract.
Keywords: 4,4′-Dianilino-1,1′-binaphthyl-5,5′disulfonic acid; 8-Anilinonaphthalene-1-sulfonic acid; ANS; CSP; Cataract; Chemical shift perturbation; Crystallin; HGS; HSQC; Human γS crystallin; Hydrophobicity; NMR spectroscopy; S–pi interactions; V41M; bis-ANS; heteronuclear single quantum coherence; the Val41Met mutant of human γS crystallin.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Increased hydrophobic surface exposure in the cataract-related G18V variant of human γS-crystallin.Biochim Biophys Acta. 2016 Jan;1860(1 Pt B):325-32. doi: 10.1016/j.bbagen.2015.09.022. Epub 2015 Oct 14. Biochim Biophys Acta. 2016. PMID: 26459004 Free PMC article.
-
Structural studies on the individual domains of human γS-crystallin and its G57W mutant unfolds mechanistic insights into childhood cataracts.Biochem Biophys Res Commun. 2019 Sep 24;517(3):499-506. doi: 10.1016/j.bbrc.2019.07.094. Epub 2019 Jul 29. Biochem Biophys Res Commun. 2019. PMID: 31371024
-
Human αB-crystallin discriminates between aggregation-prone and function-preserving variants of a client protein.Biochim Biophys Acta Gen Subj. 2020 Mar;1864(3):129502. doi: 10.1016/j.bbagen.2019.129502. Epub 2019 Dec 5. Biochim Biophys Acta Gen Subj. 2020. PMID: 31812542 Free PMC article.
-
Gamma crystallins of the human eye lens.Biochim Biophys Acta. 2016 Jan;1860(1 Pt B):333-43. doi: 10.1016/j.bbagen.2015.06.007. Epub 2015 Jun 25. Biochim Biophys Acta. 2016. PMID: 26116913 Review.
-
Genetics of crystallins: cataract and beyond.Exp Eye Res. 2009 Feb;88(2):173-89. doi: 10.1016/j.exer.2008.10.011. Epub 2008 Nov 1. Exp Eye Res. 2009. PMID: 19007775 Review.
Cited by
-
A Combined NMR and SAXS Analysis of the Partially Folded Cataract-Associated V75D γD-Crystallin.Biophys J. 2017 Mar 28;112(6):1135-1146. doi: 10.1016/j.bpj.2017.02.010. Biophys J. 2017. PMID: 28355541 Free PMC article.
-
A Novel Mutation in CRYGC Mutation Associated with Autosomal Dominant Congenital Cataracts and Microcornea.Ophthalmol Sci. 2021 Dec 17;2(1):100093. doi: 10.1016/j.xops.2021.100093. eCollection 2022 Mar. Ophthalmol Sci. 2021. PMID: 36246175 Free PMC article.
-
Mutation screening of crystallin genes in Chinese families with congenital cataracts.Mol Vis. 2019 Aug 9;25:427-437. eCollection 2019. Mol Vis. 2019. PMID: 31523120 Free PMC article.
-
Human γS-Crystallin-Copper Binding Helps Buffer against Aggregation Caused by Oxidative Damage.Biochemistry. 2020 Jun 30;59(25):2371-2385. doi: 10.1021/acs.biochem.0c00293. Epub 2020 Jun 12. Biochemistry. 2020. PMID: 32510933 Free PMC article.
-
Multiple Aggregation Pathways in Human γS-Crystallin and Its Aggregation-Prone G18V Variant.Invest Ophthalmol Vis Sci. 2017 Apr 1;58(4):2397-2405. doi: 10.1167/iovs.16-20621. Invest Ophthalmol Vis Sci. 2017. PMID: 28444328 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

