Human α1β3γ2L gamma-aminobutyric acid type A receptors: High-level production and purification in a functional state
- PMID: 24288268
- PMCID: PMC3926741
- DOI: 10.1002/pro.2401
Human α1β3γ2L gamma-aminobutyric acid type A receptors: High-level production and purification in a functional state
Abstract
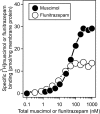
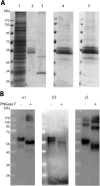
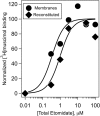
Gamma-aminobutyric acid type A receptors (GABA(A)Rs) are the most important inhibitory chloride ion channels in the central nervous system and are major targets for a wide variety of drugs. The subunit compositions of GABA(A)Rs determine their function and pharmacological profile. GABAA Rs are heteropentamers of subunits, and (α1)2 (β3)2 (γ2L)1 is a common subtype. Biochemical and biophysical studies of GABA(A)Rs require larger quantities of receptors of defined subunit composition than are currently available. We previously reported high-level production of active human α1β3 GABA(A)R using tetracycline-inducible stable HEK293 cells. Here we extend the strategy to receptors containing three different subunits. We constructed a stable tetracycline-inducible HEK293-TetR cell line expressing human (N)-FLAG-α1β3γ2L-(C)-(GGS)3 GK-1D4 GABA(A)R. These cells achieved expression levels of 70-90 pmol [(3)H]muscimol binding sites/15-cm plate at a specific activity of 15-30 pmol/mg of membrane protein. Incorporation of the γ2 subunit was confirmed by the ratio of [(3)H]flunitrazepam to [(3)H]muscimol binding sites and sensitivity of GABA-induced currents to benzodiazepines and zinc. The α1β3γ2L GABA(A)Rs were solubilized in dodecyl-D-maltoside, purified by anti-FLAG affinity chromatography and reconstituted in CHAPS/asolectin at an overall yield of ∼ 30%. Typical purifications yielded 1.0-1.5 nmoles of [(3)H]muscimol binding sites/60 plates. Receptors with similar properties could be purified by 1D4 affinity chromatography with lower overall yield. The composition of the purified, reconstituted receptors was confirmed by ligand binding, Western blot, and proteomics. Allosteric interactions between etomidate and [(3)H]muscimol binding were maintained in the purified state.
Keywords: HEK293 TetR cells; human α1β3γ2 GABAAR; purification; reconstitution.
© 2013 The Protein Society.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

