Regulation of neurotropic signaling by the inducible, NF-kB-sensitive miRNA-125b in Alzheimer's disease (AD) and in primary human neuronal-glial (HNG) cells
- PMID: 24293102
- PMCID: PMC4038663
- DOI: 10.1007/s12035-013-8595-3
Regulation of neurotropic signaling by the inducible, NF-kB-sensitive miRNA-125b in Alzheimer's disease (AD) and in primary human neuronal-glial (HNG) cells
Abstract
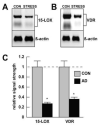
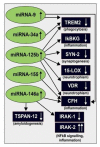
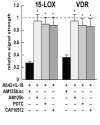
Inducible microRNAs (miRNAs) perform critical regulatory roles in central nervous system (CNS) development, aging, health, and disease. Using miRNA arrays, RNA sequencing, enhanced Northern dot blot hybridization technologies, Western immunoblot, and bioinformatics analysis, we have studied miRNA abundance and complexity in Alzheimer's disease (AD) brain tissues compared to age-matched controls. In both short post-mortem AD and in stressed primary human neuronal-glial (HNG) cells, we observe a consistent up-regulation of several brain-enriched miRNAs that are under transcriptional control by the pro-inflammatory transcription factor NF-kB. These include miRNA-9, miRNA-34a, miRNA-125b, miRNA-146a, and miRNA-155. Of the inducible miRNAs in this subfamily, miRNA-125b is among the most abundant and significantly induced miRNA species in human brain cells and tissues. Bioinformatics analysis indicated that an up-regulated miRNA-125b could potentially target the 3'untranslated region (3'-UTR) of the messenger RNA (mRNA) encoding (a) a 15-lipoxygenase (15-LOX; ALOX15; chr 17p13.3), utilized in the conversion of docosahexaneoic acid into neuroprotectin D1 (NPD1), and (b) the vitamin D3 receptor (VDR; VD3R; chr12q13.11) of the nuclear hormone receptor superfamily. 15-LOX and VDR are key neuromolecular factors essential in lipid-mediated signaling, neurotrophic support, defense against reactive oxygen and nitrogen species (reactive oxygen and nitrogen species), and neuroprotection in the CNS. Pathogenic effects appear to be mediated via specific interaction of miRNA-125b with the 3'-UTR region of the 15-LOX and VDR messenger RNAs (mRNAs). In AD hippocampal CA1 and in stressed HNG cells, 15-LOX and VDR down-regulation and a deficiency in neurotrophic support may therefore be explained by the actions of a single inducible, pro-inflammatory miRNA-125b. We will review the recent data on the pathogenic actions of this up-regulated miRNA-125b in AD and discuss potential therapeutic approaches using either anti-NF-kB or anti-miRNA-125b strategies. These may be of clinical relevance in the restoration of 15-LOX and VDR expression back to control levels and the re-establishment of homeostatic neurotrophic signaling in the CNS.
Figures

Similar articles
-
Regulation of complement factor H (CFH) by multiple miRNAs in Alzheimer's disease (AD) brain.Mol Neurobiol. 2012 Aug;46(1):11-9. doi: 10.1007/s12035-012-8234-4. Mol Neurobiol. 2012. PMID: 22302353 Free PMC article. Review.
-
NF-кB-regulated micro RNAs (miRNAs) in primary human brain cells.Exp Neurol. 2012 Jun;235(2):484-90. doi: 10.1016/j.expneurol.2011.11.022. Epub 2011 Nov 23. Exp Neurol. 2012. PMID: 22138609 Free PMC article.
-
MicroRNA (miRNA) Signaling in the Human CNS in Sporadic Alzheimer's Disease (AD)-Novel and Unique Pathological Features.Int J Mol Sci. 2015 Dec 17;16(12):30105-16. doi: 10.3390/ijms161226223. Int J Mol Sci. 2015. PMID: 26694372 Free PMC article.
-
Up-regulation of NF-kB-sensitive miRNA-125b and miRNA-146a in metal sulfate-stressed human astroglial (HAG) primary cell cultures.J Inorg Biochem. 2011 Nov;105(11):1434-7. doi: 10.1016/j.jinorgbio.2011.05.012. Epub 2011 May 27. J Inorg Biochem. 2011. PMID: 22099153 Free PMC article.
-
Lipopolysaccharides (LPSs) as Potent Neurotoxic Glycolipids in Alzheimer's Disease (AD).Int J Mol Sci. 2022 Oct 21;23(20):12671. doi: 10.3390/ijms232012671. Int J Mol Sci. 2022. PMID: 36293528 Free PMC article. Review.
Cited by
-
MicroRNAs in Alzheimer's Disease: Diagnostic Markers or Therapeutic Agents?Front Pharmacol. 2019 Jun 18;10:665. doi: 10.3389/fphar.2019.00665. eCollection 2019. Front Pharmacol. 2019. PMID: 31275145 Free PMC article. Review.
-
miR-6076 targets BCL6 in SH-SY5Y cells to regulate amyloid-β-induced neuronal damage.J Cell Mol Med. 2023 Dec;27(24):4145-4154. doi: 10.1111/jcmm.17999. Epub 2023 Oct 17. J Cell Mol Med. 2023. PMID: 37849385 Free PMC article.
-
Atsttrin reduces lipopolysaccharide-induced neuroinflammation by inhibiting the nuclear factor kappa B signaling pathway.Neural Regen Res. 2019 Nov;14(11):1994-2002. doi: 10.4103/1673-5374.259623. Neural Regen Res. 2019. PMID: 31290458 Free PMC article.
-
miR-26b inhibits total neurite outgrowth, promotes cells apoptosis and downregulates neprilysin in Alzheimer's disease.Int J Clin Exp Pathol. 2018 Jul 1;11(7):3383-3390. eCollection 2018. Int J Clin Exp Pathol. 2018. PMID: 31949715 Free PMC article.
-
The epigenetic dimension of Alzheimer's disease: causal, consequence, or curiosity?Dialogues Clin Neurosci. 2014 Sep;16(3):373-93. doi: 10.31887/DCNS.2014.16.3/mmillan. Dialogues Clin Neurosci. 2014. PMID: 25364287 Free PMC article. Review.
References
-
- Ambros V. The functions of animal microRNAs. Nature. 2004;431:350–355. - PubMed
-
- Lukiw WJ. Micro-RNA speciation in fetal, adult and Alzheimer’s disease hippocampus. Neuroreport. 2007;18:297–300. - PubMed
-
- Taft RJ, Pang KC, Mercer TR, Dinger M, Mattick JS. Non-coding RNAs: regulators of disease. J Pathol. 2010;220:126–139. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

