Canakinumab in patients with cryopyrin-associated periodic syndrome: an update for clinicians
- PMID: 24294305
- PMCID: PMC3836377
- DOI: 10.1177/1759720X13502629
Canakinumab in patients with cryopyrin-associated periodic syndrome: an update for clinicians
Abstract
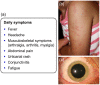
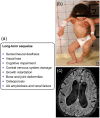
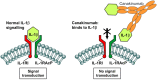
The cryopyrin-associated periodic syndrome (CAPS) is a very rare disease. It is estimated that there are 1-2 cases for every 1 million people in the US and 1 in every 360,000 in France. However, many patients are diagnosed very late or not at all, meaning the real prevalence is likely to be higher. CAPS encompasses the three entities of familial cold auto-inflammatory syndrome (FCAS), Muckle-Wells syndrome (MWS), and neonatal-onset multisystem inflammatory disease (NOMID)/chronic infantile neurologic, cutaneous and articular (CINCA) syndrome. They have in common a causative mutation in the NLRP3 gene. The altered gene product cryopyrin leads to activation of the inflammasome which in turn is responsible for excessive production of interleukin (IL)-1β. IL-1β causes the inflammatory manifestations in CAPS. These appear as systemic inflammation including fever, headache or fatigue, rash, eye disease, progressive sensorineural hearing loss, musculoskeletal manifestations and central nervous system (CNS) symptoms (NOMID/CINCA only). With the advent of IL-1 Inhibitors, safe and effective therapeutic options became available for this devastating disease. To prevent severe and possible life-threatening disease sequelae, early and correct diagnosis and immediate initiation of therapy are mandatory in most patients. Canakinumab is a fully human monoclonal IgG1 anti-IL-1β antibody. It provides selective and prolonged IL-1β blockade and has demonstrated a rapid (within hours), complete and sustained response in most CAPS patients without any consistent pattern of side effects. Long-term follow-up trials have demonstrated sustained efficacy, safety and tolerability. Canakinumab is approved by the US Food and Drug Administration for FCAS and MWS and by European Medicines Agency for treatment of all three phenotypes of CAPS.
Keywords: IL-1β-inhibition; canakinumab; cryopyrin-associated periodic syndrome (CAPS).
Conflict of interest statement
Figures


Similar articles
-
[Cryopyrin-associated periodic syndrome].Z Rheumatol. 2012 Apr;71(3):199-208. doi: 10.1007/s00393-011-0856-9. Z Rheumatol. 2012. PMID: 22527214 German.
-
Canakinumab (ACZ885, a fully human IgG1 anti-IL-1β mAb) induces sustained remission in pediatric patients with cryopyrin-associated periodic syndrome (CAPS).Arthritis Res Ther. 2011 Feb 28;13(1):R34. doi: 10.1186/ar3266. Arthritis Res Ther. 2011. PMID: 21356079 Free PMC article. Clinical Trial.
-
Long term management of patients with cryopyrin-associated periodic syndromes (CAPS): focus on rilonacept (IL-1 Trap).Biologics. 2008 Dec;2(4):733-42. doi: 10.2147/btt.s3167. Biologics. 2008. PMID: 19707454 Free PMC article.
-
CAPS--pathogenesis, presentation and treatment of an autoinflammatory disease.Semin Immunopathol. 2015 Jul;37(4):377-85. doi: 10.1007/s00281-015-0491-7. Epub 2015 May 12. Semin Immunopathol. 2015. PMID: 25963520 Review.
-
Diagnosis and Management of the Cryopyrin-Associated Periodic Syndromes (CAPS): What Do We Know Today?J Clin Med. 2021 Jan 1;10(1):128. doi: 10.3390/jcm10010128. J Clin Med. 2021. PMID: 33401496 Free PMC article. Review.
Cited by
-
Long-term safety and effectiveness of canakinumab therapy in patients with cryopyrin-associated periodic syndrome: results from the β-Confident Registry.RMD Open. 2021 May;7(2):e001663. doi: 10.1136/rmdopen-2021-001663. RMD Open. 2021. PMID: 34001647 Free PMC article.
-
[Skin ulcerations due to CINCA syndrome and its successful treatment with prostaglandin E1].Z Rheumatol. 2018 Sep;77(7):633-636. doi: 10.1007/s00393-018-0515-5. Z Rheumatol. 2018. PMID: 30066026 German.
-
IL-1β and IL-18: inflammatory markers or mediators of hypertension?Br J Pharmacol. 2014 Dec;171(24):5589-602. doi: 10.1111/bph.12876. Br J Pharmacol. 2014. PMID: 25117218 Free PMC article. Review.
-
NLRP3-inflammasomes are triggered by age-related hearing loss in the inner ear of mice.Am J Transl Res. 2017 Dec 15;9(12):5611-5618. eCollection 2017. Am J Transl Res. 2017. PMID: 29312513 Free PMC article.
-
Inflammasome activation in mouse inner ear in response to MCMV induced hearing loss.J Otol. 2015 Dec;10(4):143-149. doi: 10.1016/j.joto.2015.12.001. Epub 2016 Jan 6. J Otol. 2015. PMID: 29937798 Free PMC article.
References
-
- Aganna E., Martinon F., Hawkins P., Ross J., Swan D., Booth D., et al. (2002) Association of mutations in the NALP3/CIAS1/PYPAF1 gene with a broad phenotype including recurrent fever, cold sensitivity, sensorineural deafness, and AA amyloidosis. Arthritis Rheum 46: 2445–2452 - PubMed
-
- Aksentijevich I., Nowak M., Mallah M., Chae J., Watford W., Hofmann S., et al. (2002) De novo CIAS1 mutations, cytokine activation, and evidence for genetic heterogeneity in patients with neonatal-onset multisystem inflammatory disease (NOMID): a new member of the expanding family of pyrin-associated autoinflammatory diseases. Arthritis Rheum 46: 3340–3348 - PMC - PubMed
-
- Alten R., Gram H., Joosten L., van den Berg W., Sieper J., Wassenberg S., et al. (2008) The human anti-IL-1 beta monoclonal antibody ACZ885 is effective in joint inflammation models in mice and in a proof-of-concept study in patients with rheumatoid arthritis. Arthritis Res Ther 10(3): R67. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

