Congenital Heart Defects in Adults : A Field Guide for Cardiologists
- PMID: 24294540
- PMCID: PMC3842121
- DOI: 10.4172/2155-9880.s8-007
Congenital Heart Defects in Adults : A Field Guide for Cardiologists
Abstract
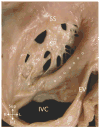
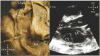
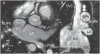
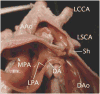
Advances in cardiology and cardiac surgery allow a large proportion of patients with congenital heart defects to survive into adulthood. These patients frequently develop complications characteristic of the defect or its treatment. Consequently, adult cardiologists participating in the care of these patients need a working knowledge of the more common defects. Occasionally, patients with congenital heart defects such as atrial septal defect, Ebstein anomaly or physiologically corrected transposition of the great arteries present for the first time in adulthood. More often patients previously treated in pediatric cardiology centers have transitioned to adult congenital heart disease centers for ongoing care. Some of the more important defects in this category are tetralogy of Fallot, transposition of the great arteries, functionally single ventricle defects, and coarctation. Through this field guide, we provide an overview of the anatomy of selected defects commonly seen in an adult congenital practice using pathology specimens and clinical imaging studies. In addition, we describe the physiology, clinical presentation to the adult cardiologist, possible complications, treatment options, and outcomes.
Figures






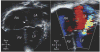




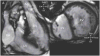



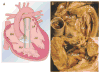



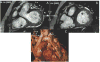
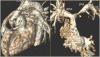

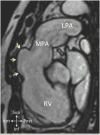
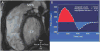





Similar articles
-
Common congenital heart defects. Long-term follow-up.Nurs Clin North Am. 1994 Jun;29(2):233-48. Nurs Clin North Am. 1994. PMID: 8202400 Review.
-
Transition of care to adult congenital heart centres: what do patients know about their heart condition?Can J Cardiol. 2002 Feb;18(2):141-6. Can J Cardiol. 2002. PMID: 11875583
-
Right Ventricular Failure and Congenital Heart Disease.Cardiol Clin. 2020 May;38(2):239-242. doi: 10.1016/j.ccl.2020.02.002. Cardiol Clin. 2020. PMID: 32284100 Review.
-
Transesophageal echocardiography (TEE) in congenital heart disease with focus on the adult.Cardiol Clin. 2000 Nov;18(4):861-92. doi: 10.1016/s0733-8651(05)70185-5. Cardiol Clin. 2000. PMID: 11236171 Review.
-
Anatomy of the ventricular septal defect in congenital heart defects: a random association?Orphanet J Rare Dis. 2018 Jul 18;13(1):118. doi: 10.1186/s13023-018-0861-z. Orphanet J Rare Dis. 2018. PMID: 30021599 Free PMC article.
Cited by
-
Paediatric/congenital cardiology physician scientists-An endangered species.Eur J Clin Invest. 2020 Oct;50(10):e13367. doi: 10.1111/eci.13367. Epub 2020 Sep 5. Eur J Clin Invest. 2020. PMID: 32735699 Free PMC article.
-
Combined spinal-epidural anesthesia for cesarean delivery in a patient with cor triloculare biventriculare.BMC Anesthesiol. 2017 Aug 29;17(1):115. doi: 10.1186/s12871-017-0411-6. BMC Anesthesiol. 2017. PMID: 28851292 Free PMC article.
References
-
- Hoffman JI, Kaplan S, Liberthson RR. Prevalence of congenital heart disease. Am Heart J. 2004;147:425–439. - PubMed
-
- Marelli AJ, Gurvitz M. From numbers to guidelines. Prog Cardiovasc Dis. 2011;53:239–246. - PubMed
-
- Lai W, Mertens L, Cohen M, Geva T. Echocardiography in Pediatric and Congenital Heart Disease: From Fetus to Adult. Wiley-Blackwell; 2009.
-
- Rosas M, Attie F. Atrial septal defect in adults. Timely Top Med Cardiovasc Dis. 2007;11:E34. - PubMed
-
- Najm HK, Williams WG, Chuaratanaphong S, Watzka SB, Coles JG, et al. Primum atrial septal defect in children: early results, risk factors, and freedom from reoperation. Ann Thorac Surg. 1998;66:829–835. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
