Resolving the roles of immunity, pathogenesis, and immigration for rabies persistence in vampire bats
- PMID: 24297874
- PMCID: PMC3870737
- DOI: 10.1073/pnas.1308817110
Resolving the roles of immunity, pathogenesis, and immigration for rabies persistence in vampire bats
Abstract
Bats are important reservoirs for emerging infectious diseases, yet the mechanisms that allow highly virulent pathogens to persist within bat populations remain obscure. In Latin America, vampire-bat-transmitted rabies virus represents a key example of how such uncertainty can impede efforts to prevent cross-species transmission. Despite decades of agricultural and human health losses, control efforts have had limited success. To establish persistence mechanisms of vampire-bat-transmitted rabies virus in Latin America, we use data from a spatially replicated, longitudinal field study of vampire bats in Peru to parameterize a series of mechanistic transmission models. We find that single-colony persistence cannot occur. Instead, dispersal of bats between colonies, combined with a high frequency of immunizing nonlethal infections, is necessary to maintain rabies virus at levels consistent with field observations. Simulations show that the strong spatial component to transmission dynamics could explain the failure of bat culls to eliminate rabies and suggests that geographic coordination of control efforts might reduce transmission to humans and domestic animals. These findings offer spatial dynamics as a mechanism for rabies persistence in bats that might be important for the understanding and control of other bat-borne pathogens.
Keywords: Desmodus rotundus; host–pathogen dynamics; spatial processes; wildlife culling; zoonotic disease.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

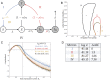
 . Demographic processes are omitted for clarity. Filled states have seroconverted, and white states are seronegative. Model IV (lifelong immunity) is a base model that includes all black lines and states. Model I (temporary immunity and lethal infection) additionally includes the blue text, model II (nonlethal infection) includes red and blue text where 1 − ρ is the probability of recovery from infection, and model III (immune boosting) includes yellow and blue where c is the strength of immune boosting that depends on the force of infection. (B) Here 95% confidence intervals (CIs) for lethal infection probability versus the effect of immigration on the force of infection. As in A, model I is colored blue, model II is colored red, model III is colored yellow, and model IV is represented by black. Models I–III arrive at the same MLE for α and ϕ (blue star), and the MLE for model IV is indicated by a black star. (C) Change in likelihood from the MLE for each model across all R0 values. Points above the horizontal gray line fall within the 95% CI (found using the likelihood ratio test), and the vertical line indicates
. Demographic processes are omitted for clarity. Filled states have seroconverted, and white states are seronegative. Model IV (lifelong immunity) is a base model that includes all black lines and states. Model I (temporary immunity and lethal infection) additionally includes the blue text, model II (nonlethal infection) includes red and blue text where 1 − ρ is the probability of recovery from infection, and model III (immune boosting) includes yellow and blue where c is the strength of immune boosting that depends on the force of infection. (B) Here 95% confidence intervals (CIs) for lethal infection probability versus the effect of immigration on the force of infection. As in A, model I is colored blue, model II is colored red, model III is colored yellow, and model IV is represented by black. Models I–III arrive at the same MLE for α and ϕ (blue star), and the MLE for model IV is indicated by a black star. (C) Change in likelihood from the MLE for each model across all R0 values. Points above the horizontal gray line fall within the 95% CI (found using the likelihood ratio test), and the vertical line indicates  . The MLE of R0 and associated 95% CIs are also shown. (D) Log likelihood (log ℒ) and ΔAIC score (with zero associated with the best-fitting model) corresponding to the MLE for each model. The ΔAIC score accounts for differences in the number of estimated parameters.
. The MLE of R0 and associated 95% CIs are also shown. (D) Log likelihood (log ℒ) and ΔAIC score (with zero associated with the best-fitting model) corresponding to the MLE for each model. The ΔAIC score accounts for differences in the number of estimated parameters.
 and
and  with 95% CIs (10−3.51, 10−2.83) and (0, 0.29), respectively, noting 0 is not included in the CI for α. Parameters for sample simulations: (A)
with 95% CIs (10−3.51, 10−2.83) and (0, 0.29), respectively, noting 0 is not included in the CI for α. Parameters for sample simulations: (A)  ,
,  ,
,  ,
,  ; (B)
; (B)  ,
,  ,
,  ,
,  ; (C)
; (C)  ,
,  ,
,  ,
,  ; and (D)
; and (D)  ,
,  ,
,  ,
,  (the MLE). In A–D, shading indicates the proportion of seropositive individuals with temporary immunity (T, light red) and the proportion of seropositive individuals that are infectious (
(the MLE). In A–D, shading indicates the proportion of seropositive individuals with temporary immunity (T, light red) and the proportion of seropositive individuals that are infectious ( , light blue). In contrast to D, simulations in A and C fail to consistently produce seropositive bats, while the dynamics in B drive the colony to extinction. At the MLE, ∼ 2 bats for every 10 susceptible bats present in a colony are expected to be exposed to VBRV by immigrant bats each year. Based on simulations this accounts for approximately half (53%) of all exposures, with the remaining exposures arising from bats within the colony. Department-specific parameters were consistent with the dynamics at the national scale (
, light blue). In contrast to D, simulations in A and C fail to consistently produce seropositive bats, while the dynamics in B drive the colony to extinction. At the MLE, ∼ 2 bats for every 10 susceptible bats present in a colony are expected to be exposed to VBRV by immigrant bats each year. Based on simulations this accounts for approximately half (53%) of all exposures, with the remaining exposures arising from bats within the colony. Department-specific parameters were consistent with the dynamics at the national scale (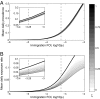
 ) and at the other, do not affect susceptible bats (lightest curve,
) and at the other, do not affect susceptible bats (lightest curve,  ). On each plot, the thick black line indicates the no-cull scenario, and the vertical gray dashed line is the MLE of ϕ; Insets magnify the results near the MLE. (A) Seroprevalence initially declines the most when susceptible bats are not culled because Eq. 1 is highest when
). On each plot, the thick black line indicates the no-cull scenario, and the vertical gray dashed line is the MLE of ϕ; Insets magnify the results near the MLE. (A) Seroprevalence initially declines the most when susceptible bats are not culled because Eq. 1 is highest when  , but in subsequent years the greatest decline corresponds to indiscriminate culling (
, but in subsequent years the greatest decline corresponds to indiscriminate culling (References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

