Randomized phase III trial of temsirolimus versus sorafenib as second-line therapy after sunitinib in patients with metastatic renal cell carcinoma
- PMID: 24297950
- PMCID: PMC5569683
- DOI: 10.1200/JCO.2013.50.3961
Randomized phase III trial of temsirolimus versus sorafenib as second-line therapy after sunitinib in patients with metastatic renal cell carcinoma
Abstract
Purpose: This international phase III trial (Investigating Torisel As Second-Line Therapy [INTORSECT]) compared the efficacy of temsirolimus (mammalian target of rapamycin inhibitor) and sorafenib (vascular endothelial growth factor receptor [VEGFR] tyrosine kinase inhibitor) as second-line therapy in patients with metastatic renal cell carcinoma (mRCC) after disease progression on sunitinib.
Patients and methods: In total, 512 patients were randomly assigned 1:1 to receive intravenous temsirolimus 25 mg once weekly (n = 259) or oral sorafenib 400 mg twice per day (n = 253), with stratification according to duration of prior sunitinib therapy (≤ or > 180 days), prognostic risk, histology (clear cell or non-clear cell), and nephrectomy status. The primary end point was progression-free survival (PFS) by independent review committee assessment. Safety, objective response rate (ORR), and overall survival (OS) were secondary end points.
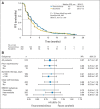
Results: Primary analysis revealed no significant difference between treatment arms for PFS (stratified hazard ratio [HR], 0.87; 95% CI, 0.71 to 1.07; two-sided P = .19) or ORR. Median PFS in the temsirolimus and sorafenib arms were 4.3 and 3.9 months, respectively. There was a significant OS difference in favor of sorafenib (stratified HR, 1.31; 95% CI, 1.05 to 1.63; two-sided P = .01). Median OS in the temsirolimus and sorafenib arms was 12.3 and 16.6 months, respectively. Safety profiles of both agents were consistent with previous studies.
Conclusion: In patients with mRCC and progression on sunitinib, second-line temsirolimus did not demonstrate a PFS advantage compared with sorafenib. The longer OS observed with sorafenib suggests sequenced VEGFR inhibition may benefit patients with mRCC.
Trial registration: ClinicalTrials.gov NCT00474786.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures


Comment in
-
Targeted therapies: Juggling combinations--not the way forward.Nat Rev Clin Oncol. 2014 Feb;11(2):64. doi: 10.1038/nrclinonc.2013.238. Epub 2013 Dec 17. Nat Rev Clin Oncol. 2014. PMID: 24343669 No abstract available.
-
Kidney cancer: Temsirolimus fails to expand its role in patients with mRCC.Nat Rev Urol. 2014 Jan;11(1):2. doi: 10.1038/nrurol.2013.308. Epub 2013 Dec 17. Nat Rev Urol. 2014. PMID: 24346004 No abstract available.
-
Maturing of renal cancer therapeutics.J Clin Oncol. 2014 Mar 10;32(8):722-4. doi: 10.1200/JCO.2013.54.1748. Epub 2014 Feb 10. J Clin Oncol. 2014. PMID: 24516015 No abstract available.
References
-
- Escudier B Eisen T Stadler WM, etal: Sorafenib in advanced clear-cell renal-cell carcinoma N Engl J Med 356:125–134,2007 - PubMed
-
- Escudier B Pluzanska A Koralewski P, etal: Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: A randomised, double-blind phase III trial Lancet 370:2103–2111,2007 - PubMed
-
- Rini BI Escudier B Tomczak P, etal: Comparative effectiveness of axitinib versus sorafenib in advanced renal cell carcinoma (AXIS): A randomised phase 3 trial Lancet 378:1931–1939,2011 - PubMed
-
- Sternberg CN Davis ID Mardiak J, etal: Pazopanib in locally advanced or metastatic renal cell carcinoma: Results of a randomized phase III trial J Clin Oncol 28:1061–1068,2010 - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

