Heterometallic triiron-oxo/hydroxo clusters: effect of redox-inactive metals
- PMID: 24304416
- PMCID: PMC3953215
- DOI: 10.1021/ja4104974
Heterometallic triiron-oxo/hydroxo clusters: effect of redox-inactive metals
Abstract
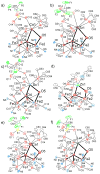
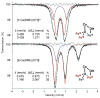
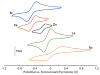
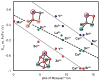
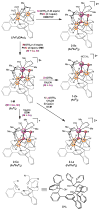
A series of tetranuclear oxo/hydroxo clusters comprised of three Fe centers and a redox-inactive metal (M) of various charge is reported. Crystallographic studies show an unprecedented Fe3M(μ4-O)(μ2-OH) core that remains intact upon changing M or the oxidation state of iron. Electrochemical studies reveal that the reduction potentials (E1/2) span a window of 500 mV and depend upon the Lewis acidity of M. Using the pKa of the M-aqua complex as a measure of Lewis acidity, these compounds display a linear dependence between E1/2 and acidity, with a slope of ∼70 mV per pKa unit. The current study of [Fe3MO(OH)] and previous ones of [Mn3MOn] (n = 2,4) moieties support the generality of the above relationship between the reduction potentials of heterometallic oxido clusters and the Lewis acidity of incorporated cations, as applied to clusters of different redox-active metals.
Figures
Similar articles
-
Redox-inactive metals modulate the reduction potential in heterometallic manganese-oxido clusters.Nat Chem. 2013 Apr;5(4):293-9. doi: 10.1038/nchem.1578. Epub 2013 Mar 3. Nat Chem. 2013. PMID: 23511417 Free PMC article.
-
Reduction potentials of heterometallic manganese-oxido cubane complexes modulated by redox-inactive metals.Proc Natl Acad Sci U S A. 2013 Jun 18;110(25):10084-8. doi: 10.1073/pnas.1302677110. Epub 2013 Jun 6. Proc Natl Acad Sci U S A. 2013. PMID: 23744039 Free PMC article.
-
Effects of Lewis Acidic Metal Ions (M) on Oxygen-Atom Transfer Reactivity of Heterometallic Mn3MO4 Cubane and Fe3MO(OH) and Mn3MO(OH) Clusters.Inorg Chem. 2019 Feb 18;58(4):2336-2345. doi: 10.1021/acs.inorgchem.8b02701. Epub 2019 Feb 7. Inorg Chem. 2019. PMID: 30730725 Free PMC article.
-
Manganese-Oxygen Intermediates in O-O Bond Activation and Hydrogen-Atom Transfer Reactions.Acc Chem Res. 2017 Nov 21;50(11):2706-2717. doi: 10.1021/acs.accounts.7b00343. Epub 2017 Oct 24. Acc Chem Res. 2017. PMID: 29064667 Review.
-
Oxidation Reactions with Bioinspired Mononuclear Non-Heme Metal-Oxo Complexes.Angew Chem Int Ed Engl. 2016 Jun 27;55(27):7632-49. doi: 10.1002/anie.201600507. Epub 2016 Jun 16. Angew Chem Int Ed Engl. 2016. PMID: 27311082 Review.
Cited by
-
Nitric oxide activation by distal redox modulation in tetranuclear iron nitrosyl complexes.J Am Chem Soc. 2015 Nov 11;137(44):14094-106. doi: 10.1021/jacs.5b07397. Epub 2015 Nov 2. J Am Chem Soc. 2015. PMID: 26390375 Free PMC article.
-
Models for Unsymmetrical Active Sites in Metalloproteins: Structural, Redox, and Magnetic Properties of Bimetallic Complexes with MII-(μ-OH)-FeIII Cores.Inorg Chem. 2017 Nov 20;56(22):14118-14128. doi: 10.1021/acs.inorgchem.7b02230. Epub 2017 Nov 7. Inorg Chem. 2017. PMID: 29112385 Free PMC article.
-
Selectivity of C-H versus C-F Bond Oxygenation by Homo- and Heterometallic Fe4 , Fe3 Mn, and Mn4 Clusters.Chemistry. 2017 Aug 10;23(45):10744-10748. doi: 10.1002/chem.201702302. Epub 2017 Jul 24. Chemistry. 2017. PMID: 28658508 Free PMC article.
-
Correlation between pH dependence of O2 evolution and sensitivity of Mn cations in the oxygen-evolving complex to exogenous reductants.Photosynth Res. 2015 Aug;125(1-2):95-103. doi: 10.1007/s11120-015-0155-4. Epub 2015 May 15. Photosynth Res. 2015. PMID: 25975707
-
Activation of a Non-Heme FeIII -OOH by a Second FeIII to Hydroxylate Strong C-H Bonds: Possible Implications for Soluble Methane Monooxygenase.Angew Chem Int Ed Engl. 2019 Jun 17;58(25):8484-8488. doi: 10.1002/anie.201903465. Epub 2019 May 9. Angew Chem Int Ed Engl. 2019. PMID: 30997707 Free PMC article.
References
-
- Fukuzumi S, Ohkubo K. Coord Chem Rev. 2010;254:372.
- Fukuzumi S. In: In Prog Inorg Chem. Karlin KD, editor. Vol. 56. John Wiley & Sons Inc; New York: 2009. p. 49.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

