Allosteric induction of the CD4-bound conformation of HIV-1 Gp120
- PMID: 24304511
- PMCID: PMC4235218
- DOI: 10.1186/1742-4690-10-147
Allosteric induction of the CD4-bound conformation of HIV-1 Gp120
Abstract
Background: HIV-1 infection of target cells is mediated via the binding of the viral envelope protein, gp120, to the cell surface receptor CD4. This interaction leads to conformational rearrangements in gp120 forming or revealing CD4 induced (CD4i) epitopes which are critical for the subsequent recognition of the co-receptor required for viral entry. The CD4-bound state of gp120 has been considered a potential immunogen for HIV-1 vaccine development. Here we report on an alternative means to induce gp120 into the CD4i conformation.
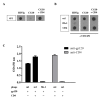
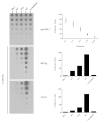
Results: Combinatorial phage display peptide libraries were screened against HIV-1 gp120 and short (14aa) peptides were selected that bind the viral envelope and allosterically induce the CD4i conformation. The lead peptide was subsequently systematically optimized for higher affinity as well as more efficient inductive activity. The peptide:gp120 complex was scrutinized with a panel of neutralizing anti-gp120 monoclonal antibodies and CD4 itself, illustrating that peptide binding does not interfere with or obscure the CD4 binding site.
Conclusions: Two surfaces of gp120 are considered targets for the development of cross neutralizing antibodies against HIV-1; the CD4 binding site and CD4i epitopes. By implementing novel peptides that allosterically induce the CD4i epitopes we have generated a viral envelope that presents both of these surfaces simultaneously.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

