Regulation of lipid accumulation by AMP-activated kinase [corrected] in high fat diet-induced kidney injury
- PMID: 24304883
- PMCID: PMC4244908
- DOI: 10.1038/ki.2013.462
Regulation of lipid accumulation by AMP-activated kinase [corrected] in high fat diet-induced kidney injury
Erratum in
- Kidney Int. 2014 Jun;85(6):1474
-
Declèves A-E, Zolkipli Z, Satriano J, et al. Regulation of lipid accumulation by AMK-activated kinase in high fat diet-induced kidney injury. Kidney Int. 2014;85:611-623.Kidney Int. 2017 Sep;92(3):769. doi: 10.1016/j.kint.2017.06.011. Kidney Int. 2017. PMID: 28807270 No abstract available.
Abstract
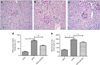
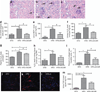
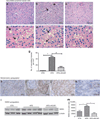
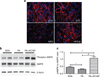
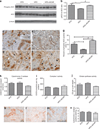
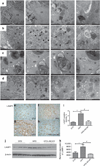
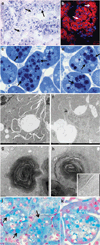
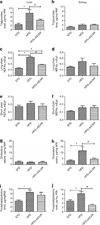
AMP-activated protein kinase (AMPK) is an important energy sensor that may be critical in regulating renal lipid accumulation. To evaluate the role of AMPK in mediating renal lipid accumulation, C57BL/6J mice were randomized to a standard diet, a high-fat diet, or a high-fat diet plus AICAR (an AMPK activator) for 14 weeks. Renal functional and structural studies along with electron microscopy were performed. Mice given the high-fat diet had proximal tubule injury with the presence of enlarged clear vacuoles, and multilaminar inclusions concurrent with an increase of tissue lipid and overloading of the lysosomal system. The margins of the clear vacuoles were positive for the endolysosomal marker, LAMP1, suggesting lysosome accumulation. Characterization of vesicles by special stains (Oil Red O, Nile Red, Luxol Fast Blue) and by electron microscopy showed they contained onion skin-like accumulations consistent with phospholipids. Moreover, cholesteryl esters and phosphatidylcholine-containing phospholipids were significantly increased in the kidneys of mice on a high-fat diet. AMPK activation with chronic AICAR treatment prevented the clinical and structural effects of high-fat diet. Thus, high-fat diet contributes to a dysfunction of the lysosomal system and altered lipid metabolism characterized by cholesterol and phospholipid accumulation in the kidney. AMPK activation normalizes the changes in renal lipid content despite chronic exposure to lipid challenge.
Figures

Similar articles
-
Whether AICAR in Pregnancy or Lactation Prevents Hypertension Programmed by High Saturated Fat Diet: A Pilot Study.Nutrients. 2020 Feb 11;12(2):448. doi: 10.3390/nu12020448. Nutrients. 2020. PMID: 32053935 Free PMC article.
-
The Adenosine Monophosphate (AMP) Analog, 5-Aminoimidazole-4-Carboxamide Ribonucleotide (AICAR) Inhibits Hepatosteatosis and Liver Tumorigenesis in a High-Fat Diet Murine Model Treated with Diethylnitrosamine (DEN).Med Sci Monit. 2018 Nov 26;24:8533-8543. doi: 10.12659/MSM.910544. Med Sci Monit. 2018. PMID: 30474622 Free PMC article.
-
Chronic AMP-activated protein kinase activation and a high-fat diet have an additive effect on mitochondria in rat skeletal muscle.J Appl Physiol (1985). 2010 Aug;109(2):511-20. doi: 10.1152/japplphysiol.00126.2010. Epub 2010 Jun 3. J Appl Physiol (1985). 2010. PMID: 20522731 Free PMC article.
-
Platycodin D inhibits lipogenesis through AMPKα-PPARγ2 in 3T3-L1 cells and modulates fat accumulation in obese mice.Planta Med. 2012 Sep;78(14):1536-42. doi: 10.1055/s-0032-1315147. Epub 2012 Aug 7. Planta Med. 2012. PMID: 22872592
-
AICAR upregulates ABCA1/ABCG1 expression in the retinal pigment epithelium and reduces Bruch's membrane lipid deposit in ApoE deficient mice.Exp Eye Res. 2021 Dec;213:108854. doi: 10.1016/j.exer.2021.108854. Epub 2021 Nov 19. Exp Eye Res. 2021. PMID: 34808137
Cited by
-
Anthocyanin-rich Seoritae extract ameliorates renal lipotoxicity via activation of AMP-activated protein kinase in diabetic mice.J Transl Med. 2015 Jun 27;13:203. doi: 10.1186/s12967-015-0563-4. J Transl Med. 2015. PMID: 26116070 Free PMC article.
-
Reprogramming of Energy Metabolism in Human PKD1 Polycystic Kidney Disease: A Systems Biology Analysis.Int J Mol Sci. 2024 Jun 29;25(13):7173. doi: 10.3390/ijms25137173. Int J Mol Sci. 2024. PMID: 39000280 Free PMC article.
-
Statins ameliorate cholesterol-induced inflammation and improve AQP2 expression by inhibiting NLRP3 activation in the kidney.Theranostics. 2020 Aug 20;10(23):10415-10433. doi: 10.7150/thno.49603. eCollection 2020. Theranostics. 2020. PMID: 32929357 Free PMC article.
-
PACS-2 deficiency in tubular cells aggravates lipid-related kidney injury in diabetic kidney disease.Mol Med. 2022 Sep 23;28(1):117. doi: 10.1186/s10020-022-00545-x. Mol Med. 2022. PMID: 36138342 Free PMC article.
-
The dipeptidyl peptidase-4 inhibitor sitagliptin protects against dyslipidemia-related kidney injury in Apolipoprotein E knockout mice.Int J Mol Sci. 2014 Jun 26;15(7):11416-34. doi: 10.3390/ijms150711416. Int J Mol Sci. 2014. PMID: 24972137 Free PMC article.
References
-
- Collins AJ, Foley RN, Herzog C, et al. US Renal Data System 2010 Annual Data Report. American journal of kidney diseases: the official journal of the National Kidney Foundation. 2011;57(1 Suppl 1):A8, e1–e526. - PubMed
-
- Marcussen N. Atubular glomeruli and the structural basis for chronic renal failure. Lab Invest. 1992;66:265–284. - PubMed
-
- Taft JL, Nolan CJ, Yeung SP, et al. Clinical and histological correlations of decline in renal function in diabetic patients with proteinuria. Diabetes. 1994;43:1046–1051. - PubMed
-
- Gilbert RE, Cooper ME. The tubulointerstitium in progressive diabetic kidney disease: more than an aftermath of glomerular injury? Kidney Int. 1999;56:1627–1637. - PubMed
-
- Chagnac A, Weinstein T, Korzets A, et al. Glomerular hemodynamics in severe obesity. Am J Physiol Renal Physiol. 2000;278:F817–F822. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

