Differential patterns of NOTCH1-4 receptor expression are markers of glioma cell differentiation
- PMID: 24305720
- PMCID: PMC3895382
- DOI: 10.1093/neuonc/not168
Differential patterns of NOTCH1-4 receptor expression are markers of glioma cell differentiation
Abstract
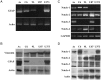
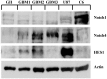
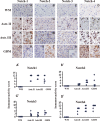
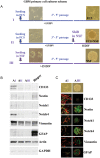
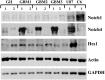
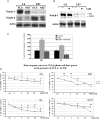
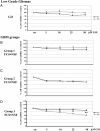
Background Notch signaling is deregulated in human gliomas and may play a role in their malignancy. However, the role of each Notch receptor in glioma cell differentiation and progression is not clear. We examined the expression pattern of Notch receptors and compared it with differentiation markers in glioma cell lines, primary human cultures, and biopsies of different grades. Furthermore, the effects of a γ-secretase inhibitor (GSI) on cell survival were assessed. Methods Notch receptors and markers of cellular differentiation were analyzed by reverse transcriptase PCR, Western blotting, immunohistochemistry, and immunocytochemistry. GSI sensitivity was assessed in both cell lines and primary cultures grown as monolayers or tumorspheres, by MTT assay. Results In cell lines, Notch1 and Notch2/4 levels paralleled those of glial fibrillary acidic protein (GFAP) and vimentin, respectively. In human gliomas and primary cultures, Notch1 was moderate/strong in low-grade tumors but weak in glioblastoma multiforme (GBM). Conversely, Notch4 increased from astrocytoma grade II to GBM. Primary GBM cultures grown in serum (monolayer) showed moderate/high levels of CD133, nestin, vimentin, and Notch4 and very low levels of GFAP and Notch1, which were reduced in tumorspheres. This effect was drastic for Notch4. GSI reduced cell survival with stronger effect in serum, whilst human primary cultures showed different sensitivity. Conclusion Data from cell lines and human gliomas suggest a correlation between expression of Notch receptors and cell differentiation. Namely, Notch1 and Notch4 are markers of differentiated and less differentiated glioma cells, respectively. We propose Notch receptors as markers of glioma grading and possible prognostic factors.
Keywords: Notch receptors; cell differentiation; gene expression; glioma cell cultures; gliomas; intermediate filaments.
Figures
References
-
- Kleihues P, Cavenee WK. World Health Organization Classification of Tumors: Pathology and Genetics of Tumors of the Nervous System. Lyon: IARC Press; 2000.
-
- Dell'Albani P. Stem cell markers in gliomas. Neurochem Res. 2008;33:2407–2415. - PubMed
-
- Fleming RJ. Structural conservation of Notch receptors and ligands. Semin Cell Dev Biol. 1998;9:599–607. - PubMed
-
- Blaumueller CM, Qi H, Zagouras P, Artavanis-Tsakonas S. Intracellular cleavage of notch leads to a heterodimeric receptor on the plasma membrane. Cell. 1997;90:281–291. - PubMed
-
- Lai EC. Notch signaling: control of cell communication and cell fate. Development. 2004;131:965–973. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

