MDR1 synonymous polymorphisms alter transporter specificity and protein stability in a stable epithelial monolayer
- PMID: 24305879
- PMCID: PMC4784985
- DOI: 10.1158/0008-5472.CAN-13-2064
MDR1 synonymous polymorphisms alter transporter specificity and protein stability in a stable epithelial monolayer
Erratum in
-
Editor's Note: MDR1 Synonymous Polymorphisms Alter Transporter Specificity and Protein Stability in a Stable Epithelial Monolayer.Cancer Res. 2025 Oct 15;85(20):4037. doi: 10.1158/0008-5472.CAN-25-3623. Cancer Res. 2025. PMID: 41088880 No abstract available.
Abstract
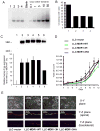
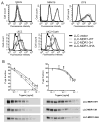
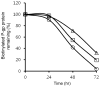
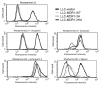
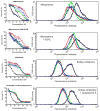
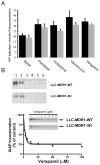
The drug efflux function of P-glycoprotein (P-gp) encoded by MDR1 can be influenced by genetic polymorphisms, including two synonymous changes in the coding region of MDR1. Here we report that the conformation of P-gp and its drug efflux activity can be altered by synonymous polymorphisms in stable epithelial monolayers expressing P-gp. Several cell lines with similar MDR1 DNA copy number were developed and termed LLC-MDR1-WT (expresses wild-type P-gp), LLC-MDR1-3H (expresses common haplotype P-gp), and LLC-MDR1-3HA (a mutant that carries a different valine codon in position 3435). These cell lines express similar levels of recombinant mRNA and protein. P-gp in each case is localized on the apical surface of polarized cells. However, the haplotype and its mutant P-gps fold differently from the wild-type, as determined by UIC2 antibody shift assays and limited proteolysis assays. Surface biotinylation experiments suggest that the non-wild-type P-gps have longer recycling times. Drug transport assays show that wild-type and haplotype P-gp respond differently to P-gp inhibitors that block efflux of rhodamine 123 or mitoxantrone. In addition, cytotoxicity assays show that the LLC-MDR1-3H cells are more resistant to mitoxantrone than the LLC-MDR1-WT cells after being treated with a P-gp inhibitor. Expression of polymorphic P-gp, however, does not affect the host cell's morphology, growth rate, or monolayer formation. Also, ATPase activity assays indicate that neither basal nor drug-stimulated ATPase activities are affected in the variant P-gps. Taken together, our findings indicate that "silent" polymorphisms significantly change P-gp function, which would be expected to affect interindividual drug disposition and response.
Conflict of interest statement
No potential conflicts of interest were disclosed.
Figures
References
-
- Schinkel AH. The physiological function of drug-transporting P-glycoproteins. Semin Cancer Biol. 1997;8:161–70. - PubMed
-
- Schinkel AH, Smit JJ, van Tellingen O, Beijnen JH, Wagenaar E, van Deemter L, et al. Disruption of the mouse mdr1a P-glycoprotein gene leads to a deficiency in the blood-brain barrier and to increased sensitivity to drugs. Cell. 1994;77:491–502. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

