Two novel proteins of cyanophage Syn5 compose its unusual horn structure
- PMID: 24307583
- PMCID: PMC3911526
- DOI: 10.1128/JVI.02479-13
Two novel proteins of cyanophage Syn5 compose its unusual horn structure
Abstract
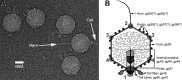
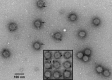
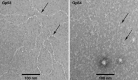
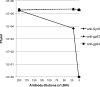
The marine cyanophage Syn5 can be propagated to a high titer in the laboratory on marine photosynthetic Synechococcus sp. strain WH8109. The purified particles carry a novel slender horn structure projecting from the vertex opposite the tail vertex. The genome of Syn5 includes a number of genes coding for novel proteins. Using immune-electron microscopy with gold-labeled antibodies, we show that two of these novel proteins, products of genes 53 and 54, are part of the horn structure. A third novel protein, the product of gene 58, is assembled onto the icosahedral capsid lattice. Characterization of radioactively labeled precursor procapsids by sucrose gradient centrifugation shows that there appear to be three classes of particles-procapsids, scaffold-deficient procapsids, and expanded capsids. These lack fully assembled horn appendages. The horn presumably assembles onto the virion just before or after DNA packaging. Antibodies raised to the recombinant novel Syn5 proteins did not interfere with phage infectivity, suggesting that the functions of these proteins are not directly involved in phage attachment or infection of the host WH8109. The horn structure may represent some adaption to the marine environment, whose function will require additional investigation.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

