Rapid detection and identification of overdose drugs in saliva by surface-enhanced Raman scattering using fused gold colloids
- PMID: 24310588
- PMCID: PMC3857074
- DOI: 10.3390/pharmaceutics3030425
Rapid detection and identification of overdose drugs in saliva by surface-enhanced Raman scattering using fused gold colloids
Abstract
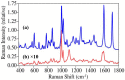
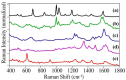
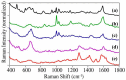
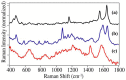
The number of drug-related emergency room visits in the United States doubled from 2004 to 2009 to 4.6 million. Consequently there is a critical need to rapidly identify the offending drug(s), so that the appropriate medical care can be administered. In an effort to meet this need we have been investigating the ability of surface-enhanced Raman spectroscopy (SERS) to detect and identify numerous drugs in saliva at ng/mL concentrations within 10 minutes. Identification is provided by matching measured spectra to a SERS library comprised of over 150 different drugs, each of which possess a unique spectrum. Trace detection is provided by fused gold colloids trapped within a porous glass matrix that generate SERS. Speed is provided by a syringe-driven sample system that uses a solid-phase extraction capillary combined with a SERS-active capillary in series. Spectral collection is provided by a portable Raman analyzer. Here we describe successful measurement of representative illicit, prescribed, and over-the-counter drugs by SERS, and 50 ng/mL cocaine in saliva as part of a focused study.
Figures




Similar articles
-
Quantitative Measurements of Codeine and Fentanyl on a Surface-Enhanced Raman-Active Pad Test.Molecules. 2019 Jul 16;24(14):2578. doi: 10.3390/molecules24142578. Molecules. 2019. PMID: 31315188 Free PMC article.
-
Rapid Analysis of Cocaine in Saliva by Surface-Enhanced Raman Spectroscopy.J Anal Bioanal Tech. 2015;6(6):1-5. doi: 10.4172/2155-9872.1000289. J Anal Bioanal Tech. 2015. PMID: 26819811 Free PMC article.
-
A Surface-Enhanced Raman Spectral Library of Important Drugs Associated With Point-of-Care and Field Applications.Front Chem. 2019 Oct 25;7:706. doi: 10.3389/fchem.2019.00706. eCollection 2019. Front Chem. 2019. PMID: 31709234 Free PMC article.
-
Development of surface-enhanced Raman spectroscopy application for determination of illicit drugs: Towards a practical sensor.Talanta. 2019 Jan 1;191:1-10. doi: 10.1016/j.talanta.2018.08.032. Epub 2018 Aug 16. Talanta. 2019. PMID: 30262036 Review.
-
Recent Advances in the Use of Surface-Enhanced Raman Scattering for Illicit Drug Detection.Sensors (Basel). 2022 May 20;22(10):3877. doi: 10.3390/s22103877. Sensors (Basel). 2022. PMID: 35632286 Free PMC article. Review.
Cited by
-
Silver nanoparticle on zinc oxide array for label-free detection of opioids through surface-enhanced raman spectroscopy.RSC Adv. 2021 Mar 17;11(19):11329-11337. doi: 10.1039/d1ra00760b. eCollection 2021 Mar 16. RSC Adv. 2021. PMID: 35423637 Free PMC article.
-
Quantitative Measurements of Codeine and Fentanyl on a Surface-Enhanced Raman-Active Pad Test.Molecules. 2019 Jul 16;24(14):2578. doi: 10.3390/molecules24142578. Molecules. 2019. PMID: 31315188 Free PMC article.
-
Rapid Detection of Amitriptyline in Dried Blood and Dried Saliva Samples with Surface-Enhanced Raman Spectroscopy.Sensors (Basel). 2022 Oct 28;22(21):8257. doi: 10.3390/s22218257. Sensors (Basel). 2022. PMID: 36365956 Free PMC article.
-
Rapid Analysis of Cocaine in Saliva by Surface-Enhanced Raman Spectroscopy.J Anal Bioanal Tech. 2015;6(6):1-5. doi: 10.4172/2155-9872.1000289. J Anal Bioanal Tech. 2015. PMID: 26819811 Free PMC article.
-
Photonics of human saliva: potential optical methods for the screening of abnormal health conditions and infections.Biophys Rev. 2021 Jun 2;13(3):359-385. doi: 10.1007/s12551-021-00807-8. eCollection 2021 Jun. Biophys Rev. 2021. PMID: 34093888 Free PMC article. Review.
References
-
- The DAWN Report. Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration; Rockville, MD, USA: Dec 28, 2010. Highlights of the 2009 Drug Abuse Warning Network (DAWN) findings on drug-related emergency department visits. Available online http://dawninfo.samhsa.gov (accessed on 26 April 2011)
-
- Rainey P.M., Schonfeld D.J. Toxicology, Monitoring of Drug Therapy, and Testing for Substance Abuse. In: Zaret B.L., Jatlow P.I., Katz L.D., editors. The Yale University School of Medicine Patient's Guide to Medical Tests. Houghton-Mifflin Co; New York, NY, USA: 1997. pp. 587–596.
-
- Burgess S. U.S. Food and Drug Administration; Silver Spring, MD, USA: Jan 13, 2011. FDA limits acetaminophen in prescription combination products; requires liver toxicity warnings. Available online http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm239894.htm (accessed on 26 April 2011)
-
- Bates D.W., Soldin S.J., Rainey P.M., Miceli J.N. Strategies for Physician Education and Therapeutic Drug Monitoring. Clin. Chem. 1998;44:401–407. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

