3-OST-7 regulates BMP-dependent cardiac contraction
- PMID: 24311987
- PMCID: PMC3849020
- DOI: 10.1371/journal.pbio.1001727
3-OST-7 regulates BMP-dependent cardiac contraction
Abstract
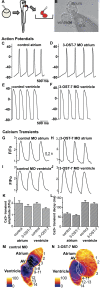
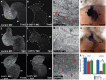
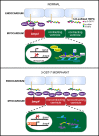
The 3-O-sulfotransferase (3-OST) family catalyzes rare modifications of glycosaminoglycan chains on heparan sulfate proteoglycans, yet their biological functions are largely unknown. Knockdown of 3-OST-7 in zebrafish uncouples cardiac ventricular contraction from normal calcium cycling and electrophysiology by reducing tropomyosin4 (tpm4) expression. Normal 3-OST-7 activity prevents the expansion of BMP signaling into ventricular myocytes, and ectopic activation of BMP mimics the ventricular noncontraction phenotype seen in 3-OST-7 depleted embryos. In 3-OST-7 morphants, ventricular contraction can be rescued by overexpression of tropomyosin tpm4 but not by troponin tnnt2, indicating that tpm4 serves as a lynchpin for ventricular sarcomere organization downstream of 3-OST-7. Contraction can be rescued by expression of 3-OST-7 in endocardium, or by genetic loss of bmp4. Strikingly, BMP misregulation seen in 3-OST-7 morphants also occurs in multiple cardiac noncontraction models, including potassium voltage-gated channel gene, kcnh2, affected in Romano-Ward syndrome and long-QT syndrome, and cardiac troponin T gene, tnnt2, affected in human cardiomyopathies. Together these results reveal 3-OST-7 as a key component of a novel pathway that constrains BMP signaling from ventricular myocytes, coordinates sarcomere assembly, and promotes cardiac contractile function.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
-
- Shi Y, Katsev S, Cai C, Evans S (2000) BMP signaling is required for heart formation in vertebrates. Dev Biol 224: 226–237. - PubMed
-
- Walters MJ, Wayman GA, Christian JL (2001) Bone morphogenetic protein function is required for terminal differentiation of the heart but not for early expression of cardiac marker genes. Mech Dev 100: 263–273. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

