Mutations in the small nuclear riboprotein 200 kDa gene (SNRNP200) cause 1.6% of autosomal dominant retinitis pigmentosa
- PMID: 24319334
- PMCID: PMC3850977
Mutations in the small nuclear riboprotein 200 kDa gene (SNRNP200) cause 1.6% of autosomal dominant retinitis pigmentosa
Abstract
Purpose: The purpose of this project was to determine the spectrum and frequency of mutations in the small nuclear riboprotein 200 kDa gene (SNRNP200) that cause autosomal dominant retinitis pigmentosa (adRP).
Methods: A well-characterized adRP cohort of 251 families was tested for mutations in the exons and intron/exon junctions of SNRNP200 using fluorescent dideoxy sequencing. An additional 21 adRP families from the eyeGENE® Network were tested for possible mutations. Bioinformatic and segregation analysis was performed on novel variants.
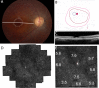
Results: SNRNP200 mutations were identified in seven of the families tested. Two previously reported mutations, p.Arg681Cys and p.Ser1087Leu, were found in two families each. One family had the previously reported p.Arg681His mutation. Two novel SNRNP200 variants, p.Pro682Ser and p.Ala542Val, were also identified in one family each. Bioinformatic and segregation analyses suggested that these novel variants are likely to be pathogenic. Clinical examination of patients with SNRNP200 mutations showed a wide range of clinical symptoms and severity, including one instance of non-penetrance.
Conclusions: Mutations in SNRNP200 caused 1.6% of disease in our adRP cohort. Pathogenic mutations were found primarily in exons 16 and 25, but the novel p.Ala542Val mutation in exon 13 suggests that variation in other genetic regions is also responsible for causing dominant disease. SNRNP200 mutations were associated with a wide range of clinical symptoms similar to those of individuals with other splice-factor gene mutations.
Figures


References
-
- Haim M. Epidemiology of retinitis pigmentosa in Denmark. Acta Ophthalmol Scand Suppl. 2002;233:1–34. - PubMed
-
- Heckenlively JR, Daiger SP. Hereditary Retinal and Choroidal Degenerations. Fifth ed: Churchill Livingston Elsevier; 2007.
-
- Kennan A, Aherne A, Humphries P. Light in retinitis pigmentosa. Trends Genet. 2005;21:103–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
