Low-oxygen culture conditions extend the multipotent properties of human retinal progenitor cells
- PMID: 24320879
- PMCID: PMC4251088
- DOI: 10.1089/ten.TEA.2013.0361
Low-oxygen culture conditions extend the multipotent properties of human retinal progenitor cells
Abstract
Purpose: Development of an effective cell-based therapy is highly dependent upon having a reproducible cell source suitable for transplantation. One potential source, isolated from the developing fetal neural retina, is the human retinal progenitor cell (hRPC). One limiting factor for the use of hRPCs is their in vitro expansion limit. As such, the aim of this study was to determine whether culturing hRPCs under 3% O2 would support their proliferative capacity while maintaining multipotency.
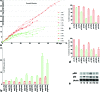
Methods: To determine the effect of low oxygen on the ability of hRPCs to self-renew, rates of proliferation and apoptosis, telomerase activity, and expression of proliferative, stemness, and differentiation markers were assessed for hRPCs cultured in 3% and 20% oxygen conditions.
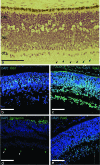
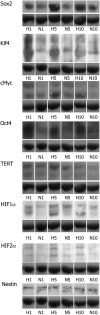
Results: Culture under 3% oxygen increases the proliferation rate and shifts the proliferation limit of hRPCs to greater 40 divisions. This increased capacity for proliferation is correlated with an upregulation of Ki67, CyclinD1, and telomerase activity and a decrease in p53 expression and apoptosis. Increased expression of cMyc, Klf4, Oct4, and Sox2 in 3% O₂ is correlated with stabilization of both HIF1α and HIF2α. The eye field development markers Pax6, Sox2, and Otx2 are present in hRPCs up to passage 16 in 3% O₂ . Following in vitro differentiation hRPCs expanded in the 3% O₂ were able to generate specialized retinal cells, including rods and cones.
Conclusions: Low-oxygen culture conditions act to maintain both multipotency and self-renewal properties of hRPCs in vitro. The extended expansion limits permit the development of a clinical-grade reagent for transplantation.
Figures


References
-
- Schmitt S.G., Aftab U., Jiang C., et al. Molecular characterization of human retinal progenitor cells. Invest Ophthalmol Vis Sci 50,5901, 2009 - PubMed
-
- Aftab U., Jiang C., Tucker B., et al. Growth kinetics and transplantation of human retinal progenitor cells. Exp Eye Res 89,301, 2009 - PubMed
-
- Hasan S.M., Vugler A.A., Miljan E.A., et al. Immortalised human foetal retinal cells retain progenitor characteristics and represent a potential source for the treatment of retinal degenerative disease. Cell Transplant 19,1291, 2010 - PubMed
-
- Marx M., Lebuhotel C., Laugier D., et al. Down regulation of pRb in cultures of avian neuroretina cells promotes proliferation of reactive Muller-like cells and emergence of retinal stem/progenitors. Exp Eye Res 90,791, 2010 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

