Remodeling and dedifferentiation of adult cardiomyocytes during disease and regeneration
- PMID: 24322910
- PMCID: PMC11113405
- DOI: 10.1007/s00018-013-1535-6
Remodeling and dedifferentiation of adult cardiomyocytes during disease and regeneration
Abstract
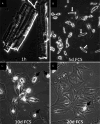
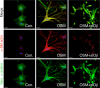
Cardiomyocytes continuously generate the contractile force to circulate blood through the body. Imbalances in contractile performance or energy supply cause adaptive responses of the heart resulting in adverse rearrangement of regular structures, which in turn might lead to heart failure. At the cellular level, cardiomyocyte remodeling includes (1) restructuring of the contractile apparatus; (2) rearrangement of the cytoskeleton; and (3) changes in energy metabolism. Dedifferentiation represents a key feature of cardiomyocyte remodeling. It is characterized by reciprocal changes in the expression pattern of "mature" and "immature" cardiomyocyte-specific genes. Dedifferentiation may enable cardiomyocytes to cope with hypoxic stress by disassembly of the energy demanding contractile machinery and by reduction of the cellular energy demand. Dedifferentiation during myocardial repair might provide cardiomyocytes with additional plasticity, enabling survival under hypoxic conditions and increasing the propensity to enter the cell cycle. Although dedifferentiation of cardiomyocytes has been described during tissue regeneration in zebrafish and newts, little is known about corresponding mechanisms and regulatory circuits in mammals. The recent finding that the cytokine oncostatin M (OSM) is pivotal for cardiomyocyte dedifferentiation and exerts strong protective effects during myocardial infarction highlights the role of cytokines as potent stimulators of cardiac remodeling. Here, we summarize the current knowledge about transient dedifferentiation of cardiomyocytes in the context of myocardial remodeling, and propose a model for the role of OSM in this process.
Conflict of interest statement
The authors declare that they have no conflicting commercial interests related to this work.
Figures

References
-
- Pohjoismaki JL, Kruger M, Al-Furoukh N, Lagerstedt A, Karhunen PJ, Braun T. Postnatal cardiomyocyte growth and mitochondrial reorganization cause multiple changes in the proteome of human cardiomyocytes. Mol BioSyst. 2013 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

