Increase in single-tablet regimen use and associated improvements in adherence-related outcomes in HIV-infected women
- PMID: 24326606
- PMCID: PMC3999284
- DOI: 10.1097/QAI.0000000000000082
Increase in single-tablet regimen use and associated improvements in adherence-related outcomes in HIV-infected women
Abstract
Introduction: The use of single-tablet antiretroviral therapy (ART) regimens and its implications on adherence among HIV-infected women have not been well described.
Methods: Participants were enrolled in the Women's Interagency HIV Study, a longitudinal study of HIV infection in US women. We examined semiannual trends in single-tablet regimen use and ART adherence, defined as self-reported 95% adherence in the past 6 months, during 2006-2013. In a nested cohort study, we assessed the comparative effectiveness of a single-tablet versus a multiple-tablet regimen with respect to adherence, virologic suppression, quality of life, and AIDS-defining events, using propensity score matching to account for demographic, behavioral, and clinical confounders. We also examined these outcomes in a subset of women switching from a multiple- to single-tablet regimen using a case-crossover design.
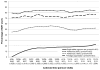
Results: We included 15,523 person-visits, representing 1727 women (53% black, 29% Hispanic, 25% IDU, median age 47). Use of single-tablet regimens among ART users increased from 7% in 2006% to 27% in 2013; adherence increased from 78% to 85% during the same period (both P < 0.001). Single-tablet regimen use was significantly associated with increased adherence (adjusted risk ratio: 1.05; 95% confidence interval: 1.03 to 1.08) and virologic suppression (risk ratio: 1.06; 95% confidence interval: 1.01 to 1.11), while associations with improved quality of life and fewer AIDS-defining events did not achieve statistical significance. Similar findings were observed among the subset of switchers.
Conclusions: Single-tablet regimen use was associated with increased adherence and virologic suppression. Despite this, 15% of women prescribed ART were still not optimally adherent; additional interventions are needed to maximize therapeutic benefits.
Conflict of interest statement
Figures
References
-
- Bangsberg DR, Perry S, Charlebois ED, Clark RA, Roberston M, Zolopa AR, et al. Non-adherence to highly active antiretroviral therapy predicts progression to AIDS. AIDS. 2001;15(9):1181–3. - PubMed
-
- Nachega JB, Hislop M, Dowdy DW, Chaisson RE, Regensberg L, Maartens G. Adherence to nonnucleoside reverse transcriptase inhibitor-based HIV therapy and virologic outcomes. Ann Intern Med. 2007;146(8):564–73. - PubMed
-
- Lazo M, Gange SJ, Wilson TE, Anastos K, Ostrow DG, Witt MD, et al. Patterns and predictors of changes in adherence to highly active antiretroviral therapy: longitudinal study of men and women. Clin Infect Dis. 2007;45(10):1377–85. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- UO1-HD-32632/HD/NICHD NIH HHS/United States
- UO1-AI-34994/AI/NIAID NIH HHS/United States
- UO1-AI-34989/AI/NIAID NIH HHS/United States
- U01 AI031834/AI/NIAID NIH HHS/United States
- U01 AI035004/AI/NIAID NIH HHS/United States
- UO1-AI-35004/AI/NIAID NIH HHS/United States
- UO1-AI-34993/AI/NIAID NIH HHS/United States
- U01 AI034994/AI/NIAID NIH HHS/United States
- P30 AI027763/AI/NIAID NIH HHS/United States
- UO1-AI-42590/AI/NIAID NIH HHS/United States
- U01 AI034993/AI/NIAID NIH HHS/United States
- UL1 RR024131/RR/NCRR NIH HHS/United States
- U01 AI034989/AI/NIAID NIH HHS/United States
- UO1-AI-31834/AI/NIAID NIH HHS/United States
- UL1 TR000004/TR/NCATS NIH HHS/United States
- U01 HD032632/HD/NICHD NIH HHS/United States
- U01 AI042590/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous