CD8+ T cells from HLA-B*57 elite suppressors effectively suppress replication of HIV-1 escape mutants
- PMID: 24330837
- PMCID: PMC3878989
- DOI: 10.1186/1742-4690-10-152
CD8+ T cells from HLA-B*57 elite suppressors effectively suppress replication of HIV-1 escape mutants
Abstract
Background: Elite Controllers or Suppressors (ES) are HIV-1 positive individuals who maintain plasma viral loads below the limit of detection of standard clinical assays without antiretroviral therapy. Multiple lines of evidence suggest that the control of viral replication in these patients is due to a strong and specific cytotoxic T lymphocyte (CTL) response. The ability of CD8+ T cells to control HIV-1 replication is believed to be impaired by the development of escape mutations. Surprisingly, viruses amplified from the plasma of ES have been shown to contain multiple escape mutations, and it is not clear how immunologic control is maintained in the face of virologic escape.
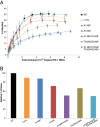
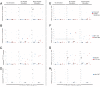
Results: We investigated the effect of escape mutations within HLA*B-57-restricted Gag epitopes on the CD8+ T cell mediated suppression of HIV-1 replication. Using site directed mutagenesis, we constructed six NL4-3 based viruses with canonical escape mutations in one to three HLA*B-57-restricted Gag epitopes. Interestingly, similar levels of CTL-mediated suppression of replication in autologous primary CD4+ T cells were observed for all of the escape mutants. Intracellular cytokine staining was performed in order to determine the mechanisms involved in the suppression of the escape variants. While low baseline CD8+ T cells responses to wild type and escape variant peptides were seen, stimulation of PBMC with either wild type or escape variant peptides resulted in increased IFN-γ and perforin expression.
Conclusions: These data presented demonstrate that CD8+ T cells from ES are capable of suppressing replication of virus harboring escape mutations in HLA-B*57-restricted Gag epitopes. Additionally, our data suggest that ES CD8+ T cells are capable of generating effective de novo responses to escape mutants.
Figures



References
-
- Deacon NJ, Tsykin A, Solomon A, Smith K, Ludford-Mending M, Hooker DJ, McPhee DA, Greenway AL, Ellett A, Chatfield C, Lawson VA, Crowe S, Maerz A, Sonza S, Learmont J, Sullivan JS, Cunningham A, Dwyer D, Dowton D, Mills J. Genomic structure of an attenuated quasi species of HIV-1 from a blood transfusion donor and recipients. Science. 1995;10:988–991. doi: 10.1126/science.270.5238.988. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

