Morphine-induced trafficking of a mu-opioid receptor interacting protein in rat locus coeruleus neurons
- PMID: 24333843
- PMCID: PMC3928604
- DOI: 10.1016/j.pnpbp.2013.12.003
Morphine-induced trafficking of a mu-opioid receptor interacting protein in rat locus coeruleus neurons
Abstract
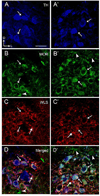
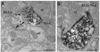
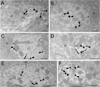
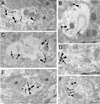
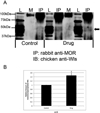
Opiate addiction is a devastating health problem, with approximately 2million people currently addicted to heroin or non-medical prescription opiates in the United States alone. In neurons, adaptations in cell signaling cascades develop following opioid actions at the mu opioid receptor (MOR). A novel putative target for intervention involves interacting proteins that may regulate trafficking of MOR. Morphine has been shown to induce a re-distribution of a MOR-interacting protein Wntless (WLS, a transport molecule necessary for secretion of neurotrophic Wnt proteins), from cytoplasmic to membrane compartments in rat striatal neurons. Given its opiate-sensitivity and its well-characterized molecular and cellular adaptations to morphine exposure, we investigated the anatomical distribution of WLS and MOR in the rat locus coeruleus (LC)-norepinephrine (NE) system. Dual immunofluorescence microscopy was used to test the hypothesis that WLS is localized to noradrenergic neurons of the LC and that WLS and MOR co-exist in common LC somatodendritic processes, providing an anatomical substrate for their putative interactions. We also hypothesized that morphine would influence WLS distribution in the LC. Rats received saline, morphine or the opiate agonist [d-Ala2, N-Me-Phe4, Gly-ol5]-enkephalin (DAMGO), and tissue sections through the LC were processed for immunogold-silver detection of WLS and MOR. Statistical analysis showed a significant re-distribution of WLS to the plasma membrane following morphine treatment in addition to an increase in the proximity of gold-silver labels for MOR and WLS. Following DAMGO treatment, MOR and WLS were predominantly localized within the cytoplasmic compartment when compared to morphine and control. In a separate cohort of rats, brains were obtained from saline-treated or heroin self-administering male rats for pulldown co-immunoprecipitation studies. Results showed an increased association of WLS and MOR following heroin exposure. As the LC-NE system is important for cognition as well as decisions underlying substance abuse, adaptations in WLS trafficking and expression may play a role in modulating MOR function in the LC and contribute to the negative sequelae of opiate exposure on executive function.
Keywords: Confocal microscopy; Electron microscopy; G-protein receptor; Norepinephrine; Trafficking; Wntless.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Opiate agonist-induced re-distribution of Wntless, a mu-opioid receptor interacting protein, in rat striatal neurons.Exp Neurol. 2012 Jan;233(1):205-13. doi: 10.1016/j.expneurol.2011.09.037. Epub 2011 Oct 6. Exp Neurol. 2012. PMID: 22001156 Free PMC article.
-
Sex differences in morphine-induced trafficking of mu-opioid and corticotropin-releasing factor receptors in locus coeruleus neurons.Brain Res. 2019 Mar 1;1706:75-85. doi: 10.1016/j.brainres.2018.11.001. Epub 2018 Nov 2. Brain Res. 2019. PMID: 30391476
-
Mu-opioid receptor redistribution in the locus coeruleus upon precipitation of withdrawal in opiate-dependent rats.Anat Rec (Hoboken). 2009 Mar;292(3):401-11. doi: 10.1002/ar.20860. Anat Rec (Hoboken). 2009. PMID: 19248160 Free PMC article.
-
Relevance of Mu-Opioid Receptor Splice Variants and Plasticity of Their Signaling Sequelae to Opioid Analgesic Tolerance.Cell Mol Neurobiol. 2021 Jul;41(5):855-862. doi: 10.1007/s10571-020-00934-y. Epub 2020 Aug 17. Cell Mol Neurobiol. 2021. PMID: 32804312 Free PMC article. Review.
-
The locus coeruleus: A key nucleus where stress and opioids intersect to mediate vulnerability to opiate abuse.Brain Res. 2010 Feb 16;1314:162-74. doi: 10.1016/j.brainres.2009.09.036. Epub 2009 Sep 16. Brain Res. 2010. PMID: 19765557 Free PMC article. Review.
Cited by
-
Addictive neurons.Ther Targets Neurol Dis. 2017;4:e1498. Epub 2017 Jan 30. Ther Targets Neurol Dis. 2017. PMID: 28649663 Free PMC article.
-
Annexin A2 traps mu-opioid receptors in recycling endosomes upon remifentanil-induced internalization.Neurobiol Pain. 2021 Aug 2;10:100071. doi: 10.1016/j.ynpai.2021.100071. eCollection 2021 Aug-Dec. Neurobiol Pain. 2021. PMID: 34401608 Free PMC article.
-
Alternative splicing of the Wnt trafficking protein, Wntless and its effects on protein-protein interactions.BMC Mol Cell Biol. 2019 Jul 8;20(1):22. doi: 10.1186/s12860-019-0208-1. BMC Mol Cell Biol. 2019. PMID: 31286866 Free PMC article.
-
Localization of the delta opioid receptor and corticotropin-releasing factor in the amygdalar complex: role in anxiety.Brain Struct Funct. 2017 Mar;222(2):1007-1026. doi: 10.1007/s00429-016-1261-6. Epub 2016 Jul 4. Brain Struct Funct. 2017. PMID: 27376372 Free PMC article.
-
Low expression of D2R and Wntless correlates with high motivation for heroin.Behav Neurosci. 2015 Dec;129(6):744-55. doi: 10.1037/bne0000104. Epub 2015 Oct 26. Behav Neurosci. 2015. PMID: 26501177 Free PMC article.
References
-
- Aghajanian GK. Tolerance of locus coeruleus neurones to morphine and suppression of withdrawal response by clonidine. Nature. 1978;276:186–188. - PubMed
-
- Aghajanian GK, Wang YY. Common alpha 2-and opiate effector mechanisms in the locus coeruleus: intracellular studies in brain slices. Neuropharmacology. 1987;26:793–799. - PubMed
-
- Arbilla S, Langer SZ. Morphine and beta-endorphin inhibit release of noradrenaline from cerebral cortex but not of dopamine from rat striatum. Nature. 1978;9:559–561. - PubMed
-
- Arden JR, Segredo V, Wang Z, Lameh J, Sadee W. Phosphorylation and agonist-specific intracellular trafficking of an epitope-tagged mu-opioid receptor expressed in HEK 293 cells. J Neurochem. 1995;65:1636–1645. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

