Ubiquity of insect-derived nitrogen transfer to plants by endophytic insect-pathogenic fungi: an additional branch of the soil nitrogen cycle
- PMID: 24334669
- PMCID: PMC3957595
- DOI: 10.1128/AEM.03338-13
Ubiquity of insect-derived nitrogen transfer to plants by endophytic insect-pathogenic fungi: an additional branch of the soil nitrogen cycle
Abstract
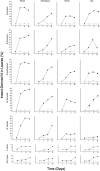
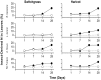
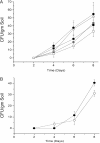
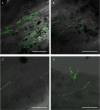
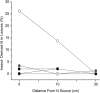
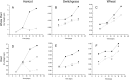
The study of symbiotic nitrogen transfer in soil has largely focused on nitrogen-fixing bacteria. Vascular plants can lose a substantial amount of their nitrogen through insect herbivory. Previously, we showed that plants were able to reacquire nitrogen from insects through a partnership with the endophytic, insect-pathogenic fungus Metarhizium robertsii. That is, the endophytic capability and insect pathogenicity of M. robertsii are coupled so that the fungus acts as a conduit to provide insect-derived nitrogen to plant hosts. Here, we assess the ubiquity of this nitrogen transfer in five Metarhizium species representing those with broad (M. robertsii, M. brunneum, and M. guizhouense) and narrower insect host ranges (M. acridum and M. flavoviride), as well as the insect-pathogenic fungi Beauveria bassiana and Lecanicillium lecanii. Insects were injected with (15)N-labeled nitrogen, and we tracked the incorporation of (15)N into two dicots, haricot bean (Phaseolus vulgaris) and soybean (Glycine max), and two monocots, switchgrass (Panicum virgatum) and wheat (Triticum aestivum), in the presence of these fungi in soil microcosms. All Metarhizium species and B. bassiana but not L. lecanii showed the capacity to transfer nitrogen to plants, although to various degrees. Endophytic association by these fungi increased overall plant productivity. We also showed that in the field, where microbial competition is potentially high, M. robertsii was able to transfer insect-derived nitrogen to plants. Metarhizium spp. and B. bassiana have a worldwide distribution with high soil abundance and may play an important role in the ecological cycling of insect nitrogen back to plant communities.
Figures
References
-
- Cocking EC. 2003. Endophytic colonization of plant roots by nitrogen-fixing bacteria. Plant Soil 252:169–175. 10.1023/A:1024106605806 - DOI
-
- Fellbauma CR, Gachomo EW, Beesetty Y, Choudharib S, Strahan GD, Pferrer PE, Toby Kiers E, Bucking H. 2012. Carbon availability triggers fungal nitrogen uptake and transport in arbuscular mycorrhizal symbiosis. Proc. Natl. Acad. Sci. U. S. A. 109:2666–2671. 10.1073/pnas.1118650109 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

