Proceedings of the 2013 National Toxicology Program Satellite Symposium
- PMID: 24334674
- PMCID: PMC3992853
- DOI: 10.1177/0192623313508020
Proceedings of the 2013 National Toxicology Program Satellite Symposium
Abstract
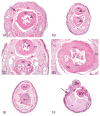
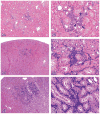
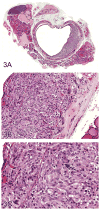
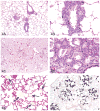
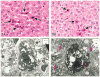
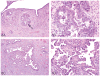
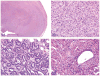
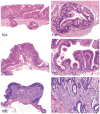
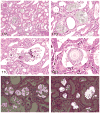
The 2013 annual National Toxicology Program (NTP) Satellite Symposium, entitled "Pathology Potpourri," was held in Portland, Oregon, in advance of the Society of Toxicologic Pathology's 32nd annual meeting. The goal of the NTP Symposium is to present current diagnostic pathology or nomenclature issues to the toxicologic pathology community. This article presents summaries of the speakers' presentations, including diagnostic or nomenclature issues that were presented, along with select images that were used for audience voting and discussion. Some lesions and topics covered during the symposium included a caudal tail vertebra duplication in mice; nephroblastematosis in rats; ectopic C cell tumor in a hamster; granular cell aggregates/tumor in the uterus of a hamster; Pneumocystis carinii in the lung of a rat; iatrogenic chronic inflammation in the lungs of control rats; hepatoblastoma arising within an adenoma in a mouse; humoral hypercalcemia of benignancy in a transgenic mouse; acetaminophen-induced hepatotoxicity in rats; electron microscopy images of iatrogenic intraerythrocytic inclusions in transgenic mice; questionable hepatocellular degeneration/cell death/artifact in rats; atypical endometrial hyperplasia in rats; malignant mixed Müllerian tumors/carcinosarcomas in rats; differential diagnoses of proliferative lesions of the intestine of rodents; and finally obstructive nephropathy caused by melamine poisoning in a rat.
Keywords: NTP Satellite Symposium; Pneumocystis carinii; atypical endometrial hyperplasia; bronchioloalveolar hyperplasia; duplicate vertebra; gastrointestinal diverticulum; granular cell aggregates; hepatoblastoma; hepatocellular apoptosis; intraerythrocytic inclusions; malignant mixed Müllerian tumor; nephroblastematosis; obstructive nephropathy..
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures









References
-
- Albers TM, Simon MA, Clifford CB. Histopathology of naturally transmitted “rat respiratory virus”: progression of lesions and proposed diagnostic criteria. Vet Pathol. 2009;46:992–9. - PubMed
-
- Ameisen JC, Idziorek T, Billaut-Mulot O, Loyens M, Tissier JP, Potentier A, Ouaissi A. Apoptosis in a unicellular eukaryote (Trypanosoma cruzi): implications for the evolutionary origin and role of programmed cell death in the control of cell proliferation, differentiation and survival. Cell Death Differ. 1995;2:285–300. - PubMed
-
- Bab I, Hajbi-Yonissi C, Gabet Y, Müller R. Micro-Tomographic Atlas of the Mouse Skeleton. Springer Science + Business Media; LLC, New York, NY: 2007. pp. 87–92.
-
- Barger AM. Erythrocyte Morphology. In: Weiss Douglas J, Jane Wardrop K., editors. Schalm's Veterinary Hematology. Blackwell Publishing; Ames, IA: 2010. pp. 144–151.
-
- Barnett SA. Genotype and environment in the tail length of mice. Q J Exp Physiol. 1965;50:417–32. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

