ER stress response mechanisms in the pathogenic yeast Candida glabrata and their roles in virulence
- PMID: 24335436
- PMCID: PMC3956515
- DOI: 10.4161/viru.27373
ER stress response mechanisms in the pathogenic yeast Candida glabrata and their roles in virulence
Abstract
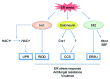
The maintenance of endoplasmic reticulum (ER) homeostasis is critical for numerous aspects of cell physiology. Eukaryotic cells respond to the accumulation of misfolded proteins in the ER (ER stress) by activating the unfolded protein response (UPR), an intracellular signaling pathway that adjusts the folding capacity of the ER. Recent studies of several pathogenic fungi have revealed that the UPR is important for antifungal resistance and virulence; therefore, the pathway has attracted much attention as a potential therapeutic target. While the UPR is highly conserved among eukaryotes, our group recently discovered that the pathogenic yeast Candida glabrata lacks the typical fungal UPR, but possesses alternative mechanisms to cope with ER stress. This review summarizes how C. glabrata responds to ER stress and discusses the impacts of ER quality control systems on antifungal resistance and virulence.
Keywords: Candida glabrata; Hac1; Ire1; Slt2; antifungal resistance; calcineurin; endoplasmic reticulum stress; regulated Ire1-dependent decay; unfolded protein response; virulence.
Figures
Similar articles
-
Dissection of Ire1 functions reveals stress response mechanisms uniquely evolved in Candida glabrata.PLoS Pathog. 2013 Jan;9(1):e1003160. doi: 10.1371/journal.ppat.1003160. Epub 2013 Jan 31. PLoS Pathog. 2013. PMID: 23382685 Free PMC article.
-
The protein kinase Ire1 impacts pathogenicity of Candida albicans by regulating homeostatic adaptation to endoplasmic reticulum stress.Cell Microbiol. 2021 May;23(5):e13307. doi: 10.1111/cmi.13307. Epub 2021 Jan 26. Cell Microbiol. 2021. PMID: 33403715 Free PMC article.
-
The unfolded protein response (UPR) pathway in Cryptococcus.Virulence. 2014 Feb 15;5(2):341-50. doi: 10.4161/viru.26774. Epub 2013 Oct 18. Virulence. 2014. PMID: 24504058 Free PMC article. Review.
-
Functional Coupling between the Unfolded Protein Response and Endoplasmic Reticulum/Golgi Ca2+-ATPases Promotes Stress Tolerance, Cell Wall Biosynthesis, and Virulence of Aspergillus fumigatus.mBio. 2020 Jun 2;11(3):e01060-20. doi: 10.1128/mBio.01060-20. mBio. 2020. PMID: 32487759 Free PMC article.
-
The Unfolded Protein Response Pathway in the Yeast Kluyveromyces lactis. A Comparative View among Yeast Species.Cells. 2018 Aug 14;7(8):106. doi: 10.3390/cells7080106. Cells. 2018. PMID: 30110882 Free PMC article. Review.
Cited by
-
Sesamol: a natural phenolic compound with promising anticandidal potential.J Pathog. 2014;2014:895193. doi: 10.1155/2014/895193. Epub 2014 Dec 9. J Pathog. 2014. PMID: 25574401 Free PMC article.
-
Endoplasmic reticulum stress and fungal pathogenesis.Fungal Biol Rev. 2014 Oct 1;28(2-3):29-35. doi: 10.1016/j.fbr.2014.07.001. Fungal Biol Rev. 2014. PMID: 25419229 Free PMC article.
-
Endoplasmic Reticulum Stress Markers and Their Possible Implications in Leprosy's Pathogenesis.Dis Markers. 2018 Dec 16;2018:7067961. doi: 10.1155/2018/7067961. eCollection 2018. Dis Markers. 2018. PMID: 30647798 Free PMC article.
-
Unmasking of CgYor1-Dependent Azole Resistance Mediated by Target of Rapamycin (TOR) and Calcineurin Signaling in Candida glabrata.mBio. 2022 Feb 22;13(1):e0354521. doi: 10.1128/mbio.03545-21. Epub 2022 Jan 18. mBio. 2022. PMID: 35038899 Free PMC article.
-
Endoplasmic reticulum stress and fungal pathogenesis converge.Virulence. 2014 Feb 15;5(2):331-3. doi: 10.4161/viru.28051. Epub 2014 Feb 6. Virulence. 2014. PMID: 24504109 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
