Specificity of the cyanobacterial orange carotenoid protein: influences of orange carotenoid protein and phycobilisome structures
- PMID: 24335507
- PMCID: PMC3912106
- DOI: 10.1104/pp.113.229997
Specificity of the cyanobacterial orange carotenoid protein: influences of orange carotenoid protein and phycobilisome structures
Abstract
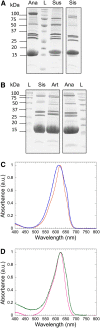
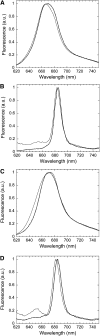
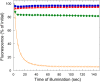
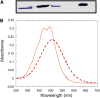
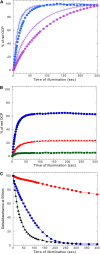
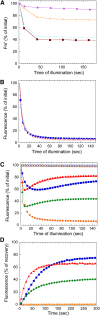
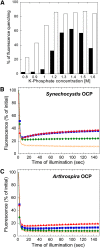
Cyanobacteria have developed a photoprotective mechanism that decreases the energy arriving at the reaction centers by increasing thermal energy dissipation at the level of the phycobilisome (PB), the extramembranous light-harvesting antenna. This mechanism is triggered by the photoactive Orange Carotenoid Protein (OCP), which acts both as the photosensor and the energy quencher. The OCP binds the core of the PB. The structure of this core differs in diverse cyanobacterial strains. Here, using two isolated OCPs and four classes of PBs, we demonstrated that differences exist between OCPs related to PB binding, photoactivity, and carotenoid binding. Synechocystis PCC 6803 (hereafter Synechocystis) OCP, but not Arthrospira platensis PCC 7345 (hereafter Arthrospira) OCP, can attach echinenone in addition to hydroxyechinenone. Arthrospira OCP binds more strongly than Synechocystis OCP to all types of PBs. Synechocystis OCP can strongly bind only its own PB in 0.8 m potassium phosphate. However, if the Synechocystis OCP binds to the PB at very high phosphate concentrations (approximately 1.4 m), it is able to quench the fluorescence of any type of PB, even those isolated from strains that lack the OCP-mediated photoprotective mechanism. Thus, the determining step for the induction of photoprotection is the binding of the OCP to PBs. Our results also indicated that the structure of PBs, at least in vitro, significantly influences OCP binding and the stabilization of OCP-PB complexes. Finally, the fact that the OCP induced large fluorescence quenching even in the two-cylinder core of Synechococcus elongatus PBs strongly suggested that OCP binds to one of the basal allophycocyanin cylinders.
Figures



Similar articles
-
In vitro reconstitution of the cyanobacterial photoprotective mechanism mediated by the Orange Carotenoid Protein in Synechocystis PCC 6803.Plant Cell. 2011 Jul;23(7):2631-43. doi: 10.1105/tpc.111.086884. Epub 2011 Jul 15. Plant Cell. 2011. PMID: 21764991 Free PMC article.
-
ApcD, ApcF and ApcE are not required for the Orange Carotenoid Protein related phycobilisome fluorescence quenching in the cyanobacterium Synechocystis PCC 6803.Biochim Biophys Acta. 2012 Aug;1817(8):1418-27. doi: 10.1016/j.bbabio.2011.11.020. Epub 2011 Dec 8. Biochim Biophys Acta. 2012. PMID: 22172739
-
Orange carotenoid protein burrows into the phycobilisome to provide photoprotection.Proc Natl Acad Sci U S A. 2016 Mar 22;113(12):E1655-62. doi: 10.1073/pnas.1523680113. Epub 2016 Mar 8. Proc Natl Acad Sci U S A. 2016. PMID: 26957606 Free PMC article.
-
The Orange Carotenoid Protein: a blue-green light photoactive protein.Photochem Photobiol Sci. 2013 Jul;12(7):1135-43. doi: 10.1039/c3pp25406b. Photochem Photobiol Sci. 2013. PMID: 23396391 Review.
-
Photoprotection in cyanobacteria: the orange carotenoid protein (OCP)-related non-photochemical-quenching mechanism.Photosynth Res. 2007 Jul-Sep;93(1-3):7-16. doi: 10.1007/s11120-007-9168-y. Epub 2007 May 8. Photosynth Res. 2007. PMID: 17486426 Review.
Cited by
-
Interdomain interactions reveal the molecular evolution of the orange carotenoid protein.Nat Plants. 2019 Oct;5(10):1076-1086. doi: 10.1038/s41477-019-0514-9. Epub 2019 Sep 16. Nat Plants. 2019. PMID: 31527845
-
Fluorescent Labeling Preserving OCP Photoactivity Reveals Its Reorganization during the Photocycle.Biophys J. 2017 Jan 10;112(1):46-56. doi: 10.1016/j.bpj.2016.11.3193. Biophys J. 2017. PMID: 28076815 Free PMC article.
-
Two-Photon-Driven Photoprotection Mechanism in Echinenone-Functionalized Orange Carotenoid Protein.J Am Chem Soc. 2025 Feb 5;147(5):4100-4110. doi: 10.1021/jacs.4c13341. Epub 2025 Jan 21. J Am Chem Soc. 2025. PMID: 39836707 Free PMC article.
-
Modulating energy arriving at photochemical reaction centers: orange carotenoid protein-related photoprotection and state transitions.Photosynth Res. 2015 Oct;126(1):3-17. doi: 10.1007/s11120-014-0031-7. Epub 2014 Aug 20. Photosynth Res. 2015. PMID: 25139327 Review.
-
Discovery of carotenoid red-shift in endolithic cyanobacteria from the Atacama Desert.Sci Rep. 2017 Sep 11;7(1):11116. doi: 10.1038/s41598-017-11581-7. Sci Rep. 2017. PMID: 28894222 Free PMC article.
References
-
- Adir N. (2005) Elucidation of the molecular structures of components of the phycobilisome: reconstructing a giant. Photosynth Res 85: 15–32 - PubMed
-
- Ajlani G, Vernotte C. (1998) Deletion of the PB-loop in the L(CM) subunit does not affect phycobilisome assembly or energy transfer functions in the cyanobacterium Synechocystis sp. PCC6714. Eur J Biochem 257: 154–159 - PubMed
-
- Ajlani G, Vernotte C, Dimagno L, Haselkorn R. (1995) Phycobilisome core mutants of Synechocystis PCC 6803. Biochim Biophys Acta 1231: 189–196
-
- Boulay C, Abasova L, Six C, Vass I, Kirilovsky D. (2008) Occurrence and function of the orange carotenoid protein in photoprotective mechanisms in various cyanobacteria. Biochim Biophys Acta 1777: 1344–1354 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

