Synthesis and structure-activity relationships of novel ecdysteroid dioxolanes as MDR modulators in cancer
- PMID: 24335576
- PMCID: PMC6269874
- DOI: 10.3390/molecules181215255
Synthesis and structure-activity relationships of novel ecdysteroid dioxolanes as MDR modulators in cancer
Abstract
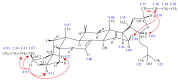
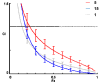
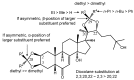
Ecdysteroids, molting hormones of insects, can exert several mild, non-hormonal bioactivities in mammals, including humans. In a previous study, we have found a significant effect of certain derivatives on the ABCB1 transporter mediated multi-drug resistance of a transfected murine leukemia cell line. In this paper, we present a structure-activity relationship study focused on the apolar dioxolane derivatives of 20-hydroxyecdysone. Semi-synthesis and bioactivity of a total of 32 ecdysteroids, including 20 new compounds, is presented, supplemented with their complete 1H- and 13C-NMR signal assignment.
Figures

Similar articles
-
Backstabbing P-gp: Side-Chain Cleaved Ecdysteroid 2,3-Dioxolanes Hyper-Sensitize MDR Cancer Cells to Doxorubicin without Efflux Inhibition.Molecules. 2017 Jan 25;22(2):199. doi: 10.3390/molecules22020199. Molecules. 2017. PMID: 28125071 Free PMC article.
-
Significant activity of ecdysteroids on the resistance to doxorubicin in mammalian cancer cells expressing the human ABCB1 transporter.J Med Chem. 2012 Jun 14;55(11):5034-43. doi: 10.1021/jm300424n. Epub 2012 May 30. J Med Chem. 2012. PMID: 22578055
-
Nitrogen-containing ecdysteroid derivatives vs. multi-drug resistance in cancer: Preparation and antitumor activity of oximes, oxime ethers and a lactam.Eur J Med Chem. 2018 Jan 20;144:730-739. doi: 10.1016/j.ejmech.2017.12.032. Epub 2017 Dec 12. Eur J Med Chem. 2018. PMID: 29291440
-
Phytoecdysteroids effects on mammalians, isolation and analysis.Mini Rev Med Chem. 2002 Jun;2(3):285-93. doi: 10.2174/1389557023406269. Mini Rev Med Chem. 2002. PMID: 12370070 Review.
-
Marine Natural Products as Models to Circumvent Multidrug Resistance.Molecules. 2016 Jul 8;21(7):892. doi: 10.3390/molecules21070892. Molecules. 2016. PMID: 27399665 Free PMC article. Review.
Cited by
-
Ecdysteroid-Containing Squalenoylated Self-Assembling Nanoparticles Exert Tumor-Selective Sensitization to Reactive Oxygen Species (ROS)-Induced Oxidative Damage While Protecting Normal Cells: Implications for Selective Radiotherapy.J Med Chem. 2025 Apr 10;68(7):7197-7212. doi: 10.1021/acs.jmedchem.4c02758. Epub 2025 Mar 28. J Med Chem. 2025. PMID: 40152540 Free PMC article.
-
Semisynthetic Ecdysteroid Cinnamate Esters and tert-Butyl Oxime Ether Derivatives with Trypanocidal Activity.J Nat Prod. 2024 Oct 25;87(10):2478-2486. doi: 10.1021/acs.jnatprod.4c00811. Epub 2024 Oct 17. J Nat Prod. 2024. PMID: 39417525 Free PMC article.
-
A Commercial Extract of Cyanotis arachnoidea Roots as a Source of Unusual Ecdysteroid Derivatives with Insect Hormone Receptor Binding Activity.J Nat Prod. 2021 Jul 23;84(7):1870-1881. doi: 10.1021/acs.jnatprod.0c01274. Epub 2021 Jun 18. J Nat Prod. 2021. PMID: 34143640 Free PMC article.
-
Ecdysteroids sensitize MDR and non-MDR cancer cell lines to doxorubicin, paclitaxel, and vincristine but tend to protect them from cisplatin.Biomed Res Int. 2015;2015:895360. doi: 10.1155/2015/895360. Epub 2015 May 17. Biomed Res Int. 2015. PMID: 26075272 Free PMC article.
-
Chemical and Biological Characterization of Metabolites from Silene viridiflora Using Mass Spectrometric and Cell-Based Assays.Biomolecules. 2024 Oct 11;14(10):1285. doi: 10.3390/biom14101285. Biomolecules. 2024. PMID: 39456218 Free PMC article.
References
-
- Karlson P. Mode of Action of Ecdysones. In: Burdette W.B., editor. Invertebrate Endocrinology and Hormonal Heterophylly. Springer; Berlin/Heidelberg, Germany: 1974. pp. 43–54.
-
- Zeleny J., Havelka J., Sláma K. Hormonally mediated insect-plant relationships: Arthropod populations associated with ecdysteroid-containing plant, Leuzea carthamoides (Asteraceae) Eur J. Entomol. 1997;94:183–198.
-
- Dinan L. A strategy for the identification of ecdysteroid receptor agonists and antagonists from plants. Eur. J. Entomol. 1995;92:271–283.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

