Internal and external influences on the rate of sensory evidence accumulation in the human brain
- PMID: 24336710
- PMCID: PMC6618757
- DOI: 10.1523/JNEUROSCI.3355-13.2013
Internal and external influences on the rate of sensory evidence accumulation in the human brain
Abstract
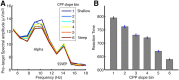
We frequently need to make timely decisions based on sensory evidence that is weak, ambiguous, or noisy resulting from conditions in the external environment (e.g., a cluttered visual scene) or within the brain itself (e.g., inattention, neural noise). Here we examine how externally and internally driven variations in the quality of sensory evidence affect the build-to-threshold dynamics of a supramodal "decision variable" signal and, hence, the timing and accuracy of decision reports in humans. Observers performed a continuous-monitoring version of the prototypical two-alternative dot-motion discrimination task, which is known to strongly benefit from sequential sampling and temporal accumulation of evidence. A centroparietal positive potential (CPP), which we previously established as a supramodal decision signal based on its invariance to motor or sensory parameters, exhibited two key identifying properties associated with the "decision variable" long described in sequential sampling models: (1) its buildup rate systematically scaled with sensory evidence strength across four levels of motion coherence, consistent with temporal integration; and (2) its amplitude reached a stereotyped level at the moment of perceptual report executions, consistent with a boundary-crossing stopping criterion. The buildup rate of the CPP also strongly predicted reaction time within coherence levels (i.e., independent of physical evidence strength), and this endogenous variation was linked with attentional fluctuations indexed by the level of parieto-occipital α-band activity preceding target onset. In tandem with the CPP, build-to-threshold dynamics were also observed in an effector-selective motor preparation signal; however, the buildup of this motor-specific process significantly lagged that of the supramodal process.
Figures



Comment in
-
An action-independent signature of perceptual choice in the human brain.J Neurosci. 2014 Apr 9;34(15):5081-2. doi: 10.1523/JNEUROSCI.0477-14.2014. J Neurosci. 2014. PMID: 24719086 Free PMC article. No abstract available.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
