Chondroitin sulfate proteoglycan tenascin-R regulates glutamate uptake by adult brain astrocytes
- PMID: 24337573
- PMCID: PMC3908396
- DOI: 10.1074/jbc.M113.504787
Chondroitin sulfate proteoglycan tenascin-R regulates glutamate uptake by adult brain astrocytes
Abstract
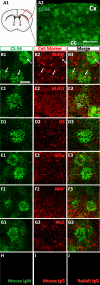
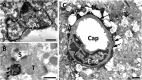
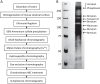
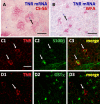
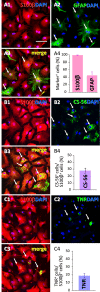
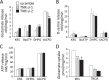
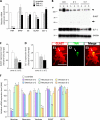
In our previous study, the CS-56 antibody, which recognizes a chondroitin sulfate moiety, labeled a subset of adult brain astrocytes, yielding a patchy extracellular matrix pattern. To explore the molecular nature of CS-56-labeled glycoproteins, we purified glycoproteins of the adult mouse cerebral cortex using a combination of anion-exchange, charge-transfer, and size-exclusion chromatographies. One of the purified proteins was identified as tenascin-R (TNR) by mass spectrometric analysis. When we compared TNR mRNA expression patterns with the distribution patterns of CS-56-positive cells, TNR mRNA was detected in CS-56-positive astrocytes. To examine the functions of TNR in astrocytes, we first confirmed that cultured astrocytes also expressed TNR protein. TNR knockdown by siRNA expression significantly reduced glutamate uptake in cultured astrocytes. Furthermore, expression of mRNA and protein of excitatory amino acid transporter 1 (GLAST), which is a major component of astrocytic glutamate transporters, was reduced by TNR knockdown. Our results suggest that TNR is expressed in a subset of astrocytes and contributes to glutamate homeostasis by regulating astrocytic GLAST expression.
Keywords: Astrocytes; Chondroitin Sulfate; Extracellular Matrix; Glutamate; Tenascin.
Figures
Similar articles
-
A review of functional heterogeneity among astrocytes and the CS56-specific antibody-mediated detection of a subpopulation of astrocytes in adult brains.Anat Sci Int. 2018 Mar;93(2):161-168. doi: 10.1007/s12565-017-0420-z. Epub 2017 Oct 31. Anat Sci Int. 2018. PMID: 29086253 Review.
-
Thrombin decreases expression of the glutamate transporter GLAST and inhibits glutamate uptake in primary cortical astrocytes via the Rho kinase pathway.Exp Neurol. 2015 Nov;273:288-300. doi: 10.1016/j.expneurol.2015.09.009. Epub 2015 Sep 21. Exp Neurol. 2015. PMID: 26391563
-
Ca(2+)-dependent reduction of glutamate aspartate transporter GLAST expression in astrocytes by P2X(7) receptor-mediated phosphoinositide 3-kinase signaling.J Neurochem. 2010 Apr;113(1):213-27. doi: 10.1111/j.1471-4159.2010.06589.x. Epub 2010 Jan 13. J Neurochem. 2010. PMID: 20070863
-
The food-associated fungal neurotoxin ochratoxin A inhibits the absorption of glutamate by astrocytes through a decrease in cell surface expression of the excitatory amino-acid transporters GLAST and GLT-1.Neurotoxicology. 2010 Sep;31(5):475-84. doi: 10.1016/j.neuro.2010.06.003. Epub 2010 Jun 15. Neurotoxicology. 2010. PMID: 20558201
-
Role of astrocytes in glutamate homeostasis: implications for excitotoxicity.Neurotox Res. 2005 Nov;8(3-4):221-5. doi: 10.1007/BF03033975. Neurotox Res. 2005. PMID: 16371316 Review.
Cited by
-
Thoracic Spinal Cord Contusion Impacts on Lumbar Enlargement: Molecular Insights.Mol Neurobiol. 2025 Jul;62(7):8551-8567. doi: 10.1007/s12035-025-04794-9. Epub 2025 Feb 27. Mol Neurobiol. 2025. PMID: 40014268
-
Microglial dyshomeostasis drives perineuronal net and synaptic loss in a CSF1R+/- mouse model of ALSP, which can be rescued via CSF1R inhibitors.Sci Adv. 2021 Aug 25;7(35):eabg1601. doi: 10.1126/sciadv.abg1601. Print 2021 Aug. Sci Adv. 2021. PMID: 34433559 Free PMC article.
-
Resolving cellular and molecular diversity along the hippocampal anterior-to-posterior axis in humans.Neuron. 2021 Jul 7;109(13):2091-2105.e6. doi: 10.1016/j.neuron.2021.05.003. Epub 2021 May 28. Neuron. 2021. PMID: 34051145 Free PMC article.
-
Extracellular matrix remodeling through endocytosis and resurfacing of Tenascin-R.Nat Commun. 2021 Dec 8;12(1):7129. doi: 10.1038/s41467-021-27462-7. Nat Commun. 2021. PMID: 34880248 Free PMC article.
-
Endogenous Optical Signals Reveal Changes of Elastin and Collagen Organization During Differentiation of Mouse Embryonic Stem Cells.Tissue Eng Part C Methods. 2015 Oct;21(10):995-1004. doi: 10.1089/ten.TEC.2014.0699. Epub 2015 Jun 17. Tissue Eng Part C Methods. 2015. PMID: 25923353 Free PMC article.
References
-
- Galtrey C. M., Fawcett J. W. (2007) The role of chondroitin sulfate proteoglycans in regeneration and plasticity in the central nervous system. Brain Res. Rev. 54, 1–18 - PubMed
-
- Carulli D., Laabs T., Geller H. M., Fawcett J. W. (2005) Chondroitin sulfate proteoglycans in neural development and regeneration. Curr. Opin. Neurobiol. 15, 116–120 - PubMed
-
- Wilson M. T., Snow D. M. (2000) Chondroitin sulfate proteoglycan expression pattern in hippocampal development: potential regulation of axon tract formation. J. Comp. Neurol. 424, 532–546 - PubMed
-
- Brückner G., Szeöke S., Pavlica S., Grosche J., Kacza J. (2006) Axon initial segment ensheathed by extracellular matrix in perineuronal nets. Neuroscience 138, 365–375 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

