Vitamin a is a negative regulator of osteoblast mineralization
- PMID: 24340023
- PMCID: PMC3858291
- DOI: 10.1371/journal.pone.0082388
Vitamin a is a negative regulator of osteoblast mineralization
Abstract
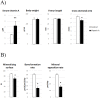
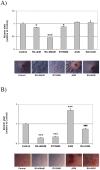
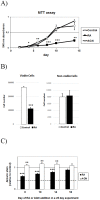
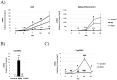
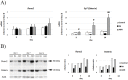
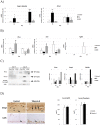
An excessive intake of vitamin A has been associated with an increased risk of fractures in humans. In animals, a high vitamin A intake leads to a reduction of long bone diameter and spontaneous fractures. Studies in rodents indicate that the bone thinning is due to increased periosteal bone resorption and reduced radial growth. Whether the latter is a consequence of direct effects on bone or indirect effects on appetite and general growth is unknown. In this study we therefore used pair-feeding and dynamic histomorphometry to investigate the direct effect of a high intake of vitamin A on bone formation in rats. Although there were no differences in body weight or femur length compared to controls, there was an approximately halved bone formation and mineral apposition rate at the femur diaphysis of rats fed vitamin A. To try to clarify the mechanism(s) behind this reduction, we treated primary human osteoblasts and a murine preosteoblastic cell line (MC3T3-E1) with the active metabolite of vitamin A; retinoic acid (RA), a retinoic acid receptor (RAR) antagonist (AGN194310), and a Cyp26 inhibitor (R115866) which blocks endogenous RA catabolism. We found that RA, via RARs, suppressed in vitro mineralization. This was independent of a negative effect on osteoblast proliferation. Alkaline phosphatase and bone gamma carboxyglutamate protein (Bglap, Osteocalcin) were drastically reduced in RA treated cells and RA also reduced the protein levels of Runx2 and Osterix, key transcription factors for progression to a mature osteoblast. Normal osteoblast differentiation involved up regulation of Cyp26b1, the major enzyme responsible for RA degradation, suggesting that a drop in RA signaling is required for osteogenesis analogous to what has been found for chondrogenesis. In addition, RA decreased Phex, an osteoblast/osteocyte protein necessary for mineralization. Taken together, our data indicate that vitamin A is a negative regulator of osteoblast mineralization.
Conflict of interest statement
Figures
References
-
- Melhus H, Michaëlsson K, Kindmark A, Bergström R, Holmberg L, et al. (1998) Excessive dietary intake of vitamin A is associated with reduced bone mineral density and increased risk for hip fracture. Ann Intern Med 129: 770–778. - PubMed
-
- Michaëlsson K, Lithell H, Vessby B, Melhus H (2003) Serum retinol levels and the risk of fracture. N Engl J Med 348: 287–294. - PubMed
-
- Feskanich D, Singh V, Willett WC, Colditz GA (2002) Vitamin A intake and hip fractures among postmenopausal women. JAMA 287: 47–54. - PubMed
-
- Binkley N, Krueger D (2000) Hypervitaminosis A and bone. Nutr Rev 58: 138–144. - PubMed
-
- Johansson S, Lind PM, Hakansson H, Oxlund H, Orberg J, et al. (2002) Subclinical hypervitaminosis A causes fragile bones in rats. Bone 31: 685–689. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

