Ventriculomegaly associated with ependymal gliosis and declines in barrier integrity in the aging human and mouse brain
- PMID: 24341850
- PMCID: PMC3954884
- DOI: 10.1111/acel.12184
Ventriculomegaly associated with ependymal gliosis and declines in barrier integrity in the aging human and mouse brain
Abstract
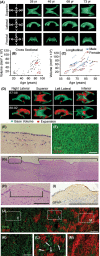
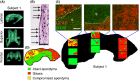
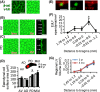
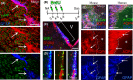
Age-associated ventriculomegaly is typically attributed to neurodegeneration; however, additional factors might initiate or contribute to progressive ventricular expansion. By directly linking postmortem human MRI sequences with histological features of periventricular tissue, we show that substantial lateral ventricle surface gliosis is associated with ventriculomegaly. To examine whether loss of ependymal cell coverage resulting in ventricle surface glial scarring can lead directly to ventricle enlargement independent of any other injury or degenerative loss, we modeled in mice the glial scarring found along the lateral ventricle surface in aged humans. Neuraminidase, which cleaves glycosidic linkages of apical adherens junction proteins, was administered intracerebroventricularly to denude areas of ependymal cells. Substantial ependymal cell loss resulted in reactive gliosis rather than stem cell-mediated regenerative repair of the ventricle lining, and the gliotic regions showed morphologic and phenotypic characteristics similar to those found in aged humans. Increased levels of aquaporin-4, indicative of edema, observed in regions of periventricular gliosis in human tissue were also replicated in our mouse model. 3D modeling together with volume measurements revealed that mice with ventricle surface scarring developed expanded ventricles, independent of neurodegeneration. Through a comprehensive, comparative analysis of the lateral ventricles and associated periventricular tissue in aged humans and mouse, followed by modeling of surface gliosis in mice, we have demonstrated a direct link between lateral ventricle surface gliosis and ventricle enlargement. These studies highlight the importance of maintaining an intact ependymal cell lining throughout aging.
Keywords: aging; ependymal cells; human; injury; lateral ventricle; mouse; neural stem cells; ventriculomegaly.
© 2013 The Authors. Aging Cell published by the Anatomical Society and John Wiley & Sons Ltd.
Figures


References
-
- Bergmann O, Liebl J, Bernard S, Alkass K, Yeung MS, Steier P, Kutschera W, Johnson L, Landen M, Druid H, Spalding KL, Frisen J. The age of olfactory bulb neurons in humans. Neuron. 2012;74:634–639. - PubMed
-
- Bouab M, Paliouras GN, Aumont A, Forest-Berard K, Fernandes KJ. Aging of the subventricular zone neural stem cell niche: evidence for quiescence-associated changes between early and mid-adulthood. Neuroscience. 2011;173:135–149. - PubMed
-
- Conover JC, Doetsch F, Garcia-Verdugo JM, Gale NW, Yancopoulos GD, Alvarez-Buylla A. Disruption of Eph/ephrin signaling affects migration and proliferation in the adult subventricular zone. Nat. Neurosci. 2000;3:1091–1097. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

