Reversal of multidrug resistance by 5,5'-dimethoxylariciresinol-4-O-β-D-glucoside in doxorubicin-resistant human leukemia K562/DOX
- PMID: 24347768
- PMCID: PMC3847250
- DOI: 10.4103/0253-7613.121371
Reversal of multidrug resistance by 5,5'-dimethoxylariciresinol-4-O-β-D-glucoside in doxorubicin-resistant human leukemia K562/DOX
Abstract
Objective: The objective of this study was to investigate the reversal effects of 5,5'-dimethoxylariciresinol-4'-O-β-D-glucoside (DMAG) extracted from traditional Chinese medicines Mahonia on multidrug resistance (MDR) of human leukemia cells to chemotherapeutic agents.
Materials and methods: MTT(3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was performed to determine the effect of DMAG on doxorubicin sensitivity to K562/DOX cells. Propidium iodide /Hoechst 33342 double staining assay was used to investigate the effect of DMAG on doxorubicin-induced cellular apoptosis. Intracellular accumulation of doxorubicin and rhodamine 123 assay were performed to evaluate the effect of DMAG on drugs efflux activity of P-glycoprotein.
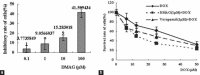
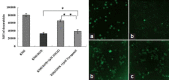
Results: DMAG significantly enhanced the doxorubicin cytotoxicity to K562/DOX cells. In the presence of 1.0 μM of DMAG, the IC50 of doxorubicin decreased from 34.93 ± 1.37 μM to 12.51 ± 1.28 μM. DMAG of 1.0 μM significantly enhanced doxorubicin-induced cell apoptosis in K562/DOX cells and the enhancement was time-dependent. A significant increase in accumulation of doxorubicin in the presence of DMAG was observed. After treatment of the K562/DOX cells for 1 h with 15.0 μM doxorubicin alone, the fluorescence intensity was 33093.12. With the addition of 1.0 μM of DMAG, the fluorescence intensity of doxorubicin was 2.3-fold higher. A significant increase of accumulation of rhodamine 123 in the presence of DMAG was also observed. With the addition of 1.0 μM of DMAG, the fluorescence intensity was increased by 49.11% compared with rhodamine 123 alone.
Conclusion: DMAG was shown to effectively enhance chemosensitivity of resistant cells, which makes it might be a suitable candidate for potential MDR-reversing agents.
Keywords: 5,5’-dimethoxylariciresinol-4’-O-β-D-glucoside; doxorubicin; leukemia; multidrug resistance.
Conflict of interest statement
Figures


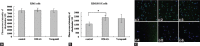
Similar articles
-
Reversal of P-glycoprotein-mediated multidrug resistance by guggulsterone in doxorubicin-resistant human myelogenous leukemia (K562/DOX) cells.Pharmazie. 2009 Oct;64(10):660-5. Pharmazie. 2009. PMID: 19947169
-
Reversal of p-glycoprotein-mediated multidrug resistance by CJX1, an amlodipine derivative, in doxorubicin-resistant human myelogenous leukemia (K562/DOX) cells.Life Sci. 2005 Sep 16;77(18):2221-32. doi: 10.1016/j.lfs.2004.12.050. Life Sci. 2005. PMID: 15967469
-
The synergistic reversal effect of multidrug resistance by quercetin and hyperthermia in doxorubicin-resistant human myelogenous leukemia cells.Int J Hyperthermia. 2008 Mar;24(2):151-9. doi: 10.1080/02656730701843109. Int J Hyperthermia. 2008. PMID: 18283591
-
Reversal effect of isotetrandrine, an isoquinoline alkaloid extracted from Caulis Mahoniae, on P-glycoprotein-mediated doxorubicin-resistance in human breast cancer (MCF-7/DOX) cells.Yao Xue Xue Bao. 2008 May;43(5):461-6. Yao Xue Xue Bao. 2008. PMID: 18717331
-
Reversal of multidrug resistance by transferrin-conjugated liposomes co-encapsulating doxorubicin and verapamil.J Pharm Pharm Sci. 2007;10(3):350-7. J Pharm Pharm Sci. 2007. PMID: 17727798
Cited by
-
Efficacy of doxorubicin-transferrin conjugate in apoptosis induction in human leukemia cells through reactive oxygen species generation.Cell Oncol (Dordr). 2016 Apr;39(2):107-18. doi: 10.1007/s13402-015-0256-2. Epub 2015 Nov 26. Cell Oncol (Dordr). 2016. PMID: 26611752 Free PMC article.
-
Hypoxia promotes chemoresistance in acute lymphoblastic leukemia cell lines by modulating death signaling pathways.BMC Cancer. 2016 Sep 22;16(1):746. doi: 10.1186/s12885-016-2776-1. BMC Cancer. 2016. PMID: 27658583 Free PMC article.
References
-
- Longley DB, Johnston PG. Molecular mechanisms of drug resistance. J Pathol. 2005;205:275–92. - PubMed
-
- Tiwari AK, Sodani K, Chen ZS. Current advances in modulation of ABC transporter-mediated multidrug resistance in cancer. Int J Toxicol Pharmacol Res. 2009;1:1–6.
-
- Ambudkar SV, Dey S, Hrycyna CA, Ramachandra M, Pastan I, Gottesman MM. Biochemical, cellular, and pharmacological aspects of the multidrug transporter. Annu Rev Pharmacol Toxicol. 1999;39:361–98. - PubMed
-
- Ling V. Multidrug resistance: Molecular mechanisms and clinical relevance. Cancer Chemother Pharmacol. 1997;40(Suppl):S3–8. - PubMed
-
- Perez EA. Impact, mechanisms, and novel chemotherapy strategies for overcoming resistance to anthracyclines and taxanes in metastatic breast cancer. Breast Cancer Res Treat. 2009;114:195–201. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

