Insulin protects apoptotic cardiomyocytes from hypoxia/reoxygenation injury through the sphingosine kinase/sphingosine 1-phosphate axis
- PMID: 24349009
- PMCID: PMC3859498
- DOI: 10.1371/journal.pone.0080644
Insulin protects apoptotic cardiomyocytes from hypoxia/reoxygenation injury through the sphingosine kinase/sphingosine 1-phosphate axis
Abstract
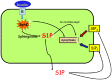
Objective: Experimental and clinical studies have shown that administration of insulin during reperfusion is cardioprotective, but the mechanisms underlying this effect are still unknown. In this study, the ability of insulin to protect apoptotic cardiomyocytes from hypoxia/reoxygenation injury using the sphingosine kinase/sphingosine 1-phosphate axis was investigated.
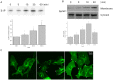
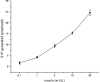
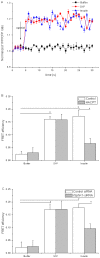
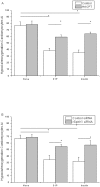
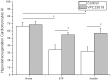
Methods and results: Rat cardiomyocytes were isolated and subjected to hypoxia and reoxygenation. [γ-32P] ATP was used to assess sphingosine kinase activity. Insulin was found to increase sphingosine kinase activity. Immunocytochemistry and Western blot analysis showed changes in the subcellular location of sphingosine kinase 1 from cytosol to the membrane in cardiomyocytes. Insulin caused cardiomyocytes to accumulate of S1P in a dose-dependent manner. FRET efficiency showed that insulin also transactivates the S1P1 receptor. TUNEL staining showed that administration of insulin during reoxygenation could to reduce the rate of reoxygenation-induced apoptosis, which is a requirement for SphK 1 activity. It also reduced the rate of activation of the S1P receptor and inhibited hypoxia/reoxygenation-induced cell death in cardiomyocytes.
Conclusion: The sphingosine kinase 1/sphingosine 1-phosphate/S1P receptor axis is one pathway through which insulin protects rat cardiomyocytes from apoptosis induced by hypoxia/reoxygenation injury.
Conflict of interest statement
Figures

Similar articles
-
[Role and related mechanism of S1P/S1P1 signal pathway during post conditioning of hypertrophic cardiomyocytes].Zhonghua Xin Xue Guan Bing Za Zhi. 2016 May 24;44(5):431-5. doi: 10.3760/cma.j.issn.0253-3758.2016.05.013. Zhonghua Xin Xue Guan Bing Za Zhi. 2016. PMID: 27220580 Chinese.
-
Sphingosine kinase and sphingosine 1-phosphate in cardioprotection.J Cardiovasc Pharmacol. 2009 Mar;53(3):189-97. doi: 10.1097/FJC.0b013e3181926706. J Cardiovasc Pharmacol. 2009. PMID: 19247197 Free PMC article. Review.
-
Protective effects of sphingosine-1-phosphate receptor agonist treatment after myocardial ischaemia-reperfusion.Cardiovasc Res. 2009 Jul 15;83(2):285-93. doi: 10.1093/cvr/cvp137. Epub 2009 May 5. Cardiovasc Res. 2009. PMID: 19416991
-
Sphingosine 1-phosphate S1P2 and S1P3 receptor-mediated Akt activation protects against in vivo myocardial ischemia-reperfusion injury.Am J Physiol Heart Circ Physiol. 2007 Jun;292(6):H2944-51. doi: 10.1152/ajpheart.01331.2006. Epub 2007 Feb 9. Am J Physiol Heart Circ Physiol. 2007. PMID: 17293497
-
Sphingosine kinase and sphingosine 1-phosphate in the heart: a decade of progress.Biochim Biophys Acta. 2013 Jan;1831(1):203-12. doi: 10.1016/j.bbalip.2012.06.006. Epub 2012 Jun 23. Biochim Biophys Acta. 2013. PMID: 22735359 Free PMC article. Review.
Cited by
-
Berberine protects HK-2 cells from hypoxia/reoxygenation induced apoptosis via inhibiting SPHK1 expression.J Nat Med. 2018 Mar;72(2):390-398. doi: 10.1007/s11418-017-1152-z. Epub 2017 Dec 19. J Nat Med. 2018. PMID: 29260413
-
Glucocorticoid Inhibition of Estrogen Regulation of the Serotonin Receptor 2B in Cardiomyocytes Exacerbates Cell Death in Hypoxia/Reoxygenation Injury.J Am Heart Assoc. 2021 Sep 7;10(17):e015868. doi: 10.1161/JAHA.120.015868. Epub 2021 Sep 2. J Am Heart Assoc. 2021. PMID: 34472367 Free PMC article.
-
Study on the Antihypertensive Mechanism of Astragalus membranaceus and Salvia miltiorrhiza Based on Intestinal Flora-Host Metabolism.Evid Based Complement Alternat Med. 2019 Jul 21;2019:5418796. doi: 10.1155/2019/5418796. eCollection 2019. Evid Based Complement Alternat Med. 2019. PMID: 31428172 Free PMC article.
-
Implication of sphingosin-1-phosphate in cardiovascular regulation.Front Biosci (Landmark Ed). 2016 Jun 1;21(7):1296-313. doi: 10.2741/4458. Front Biosci (Landmark Ed). 2016. PMID: 27100508 Free PMC article. Review.
References
-
- Piper HM, Abdallah Y, Schafer C (2004) The first minutes of reperfusion: a window of opportunity for cardioprotection. Cardiovasc Res 61: 365–371. - PubMed
-
- Yellon DM, Hausenloy DJ (2007) Myocardial reperfusion injury. N Engl J Med 357: 1121–1135. - PubMed
-
- Sodi-Pallares D, Testelli MR, Fishleder BL, Bisteni A, Medrano GA, et al. (1962) Effects of an intravenous infusion of a potassium-glucose-insulin solution on the electrocardiographic signs of myocardial infarction. A preliminary clinical report. Am J Cardiol 9: 166–181. - PubMed
-
- Jonassen AK, Brar BK, Mjos OD, Sack MN, Latchman DS, et al. (2000) Insulin administered at reoxygenation exerts a cardioprotective effect in myocytes by a possible anti-apoptotic mechanism. J Mol Cell Cardiol 32: 757–764. - PubMed
-
- Gao F, Tao L, Yan W, Gao E, Liu HR, et al. (2004) Early anti-apoptosis treatment reduces myocardial infarct size after a prolonged reperfusion. Apoptosis 9: 553–559. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

