The microenvironment-specific transformation of adult stem cells models malignant triton tumors
- PMID: 24349213
- PMCID: PMC3857244
- DOI: 10.1371/journal.pone.0082173
The microenvironment-specific transformation of adult stem cells models malignant triton tumors
Abstract
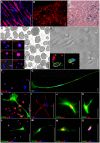
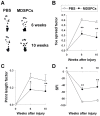
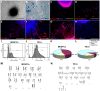
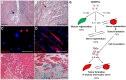
Here, we demonstrated the differentiation potential of murine muscle-derived stem/progenitor cells (MDSPCs) toward myogenic, neuronal, and glial lineages. MDSPCs, following transplantation into a critical-sized sciatic nerve defect in mice, showed full regeneration with complete functional recovery of the injured peripheral nerve at 6 weeks post-implantation. However, several weeks after regeneration of the sciatic nerve, neoplastic growths were observed. The resulting tumors were malignant peripheral nerve sheath tumors (MPNSTs) with rhabdomyoblastic differentiation, expressing myogenic, neurogenic, and glial markers, common markers of human malignant triton tumors (MTTs). No signs of tumorigenesis were observed 17 weeks post-implantation of MDSPCs into the gastrocnemius muscles of dystrophic/mdx mice, or 1 year following subcutaneous or intravenous injection. While MDSPCs were not oncogenic in nature, the neoplasias were composed almost entirely of donor cells. Furthermore, cells isolated from the tumors were serially transplantable, generating tumors when reimplanted into mice. However, this transformation could be abrogated by differentiation of the cells toward the neurogenic lineage prior to implantation. These results establish that MDSPCs participated in the regeneration of the injured peripheral nerve but transformed in a microenvironment- and time-dependent manner, when they likely received concomitant neurogenic and myogenic differentiation signals. This microenvironment-specific transformation provides a useful mouse model for human MTTs and potentially some insight into the origins of this disease.
Conflict of interest statement
Figures


Similar articles
-
Human muscle-derived stem/progenitor cells promote functional murine peripheral nerve regeneration.J Clin Invest. 2014 Apr;124(4):1745-56. doi: 10.1172/JCI44071. Epub 2014 Mar 18. J Clin Invest. 2014. PMID: 24642464 Free PMC article.
-
Malignant transformation of multipotent muscle-derived cells by concurrent differentiation signals.Stem Cells. 2007 Sep;25(9):2302-11. doi: 10.1634/stemcells.2006-0773. Epub 2007 Jun 14. Stem Cells. 2007. PMID: 17569791
-
Human adipose-derived mesenchymal stem cells systemically injected promote peripheral nerve regeneration in the mouse model of sciatic crush.Tissue Eng Part A. 2012 Jun;18(11-12):1264-72. doi: 10.1089/ten.TEA.2011.0491. Epub 2012 Apr 3. Tissue Eng Part A. 2012. PMID: 22332955
-
Regulation of stemness and stem cell niche of mesenchymal stem cells: implications in tumorigenesis and metastasis.J Cell Physiol. 2010 Feb;222(2):268-77. doi: 10.1002/jcp.21940. J Cell Physiol. 2010. PMID: 19847802 Review.
-
Stem cells treatment for sciatic nerve injury.Expert Opin Biol Ther. 2011 Dec;11(12):1591-7. doi: 10.1517/14712598.2011.628933. Epub 2011 Oct 13. Expert Opin Biol Ther. 2011. PMID: 21995439 Review.
Cited by
-
The importance of surgical margins in malignant Triton tumour of the trunk and extremities.Oncol Lett. 2021 Feb;21(2):120. doi: 10.3892/ol.2020.12381. Epub 2020 Dec 15. Oncol Lett. 2021. PMID: 33376551 Free PMC article.
-
Injured Nerve Regeneration using Cell-Based Therapies: Current Challenges.Acta Naturae. 2015 Jul-Sep;7(3):38-47. Acta Naturae. 2015. PMID: 26483958 Free PMC article.
-
Factors Within the Endoneurial Microenvironment Act to Suppress Tumorigenesis of MPNST.Front Cell Neurosci. 2018 Oct 11;12:356. doi: 10.3389/fncel.2018.00356. eCollection 2018. Front Cell Neurosci. 2018. PMID: 30364248 Free PMC article.
References
-
- Powell K (2005) Stem-cell niches: it's the ecology, stupid! Nature. 435: 268–270. - PubMed
-
- Blau HM, Brazelton TR, Weimann JM (2001) The evolving concept of a stem cell: entity or function? Cell 105: 829–841. - PubMed
-
- Rizo A, Vellenga E, de Haan G, Schuringa JJ (2006) Signaling pathways in self-renewing hematopoietic and leukemic stem cells: do all stem cells need a niche? Hum Mol Genet 15 Spec No 2: R210–219. - PubMed
-
- Doetsch F (2003) A niche for adult neural stem cells. Curr Opin Genet Dev 13: 543–550. - PubMed
-
- Cao Q, Benton RL, Whittemore SR (2002) Stem cell repair of central nervous system injury. J Neurosci Res 68: 501–510. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

