Dynamics of defense responses and cell fate change during Arabidopsis-Pseudomonas syringae interactions
- PMID: 24349466
- PMCID: PMC3859648
- DOI: 10.1371/journal.pone.0083219
Dynamics of defense responses and cell fate change during Arabidopsis-Pseudomonas syringae interactions
Abstract
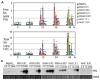
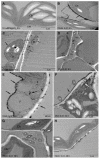
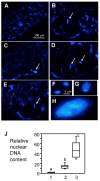
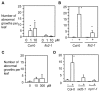
Plant-pathogen interactions involve sophisticated action and counteraction strategies from both parties. Plants can recognize pathogen derived molecules, such as conserved pathogen associated molecular patterns (PAMPs) and effector proteins, and subsequently activate PAMP-triggered immunity (PTI) and effector-triggered immunity (ETI), respectively. However, pathogens can evade such recognitions and suppress host immunity with effectors, causing effector-triggered susceptibility (ETS). The differences among PTI, ETS, and ETI have not been completely understood. Toward a better understanding of PTI, ETS, and ETI, we systematically examined various defense-related phenotypes of Arabidopsis infected with different Pseudomonas syringae pv. maculicola ES4326 strains, using the virulence strain DG3 to induce ETS, the avirulence strain DG34 that expresses avrRpm1 (recognized by the resistance protein RPM1) to induce ETI, and HrcC(-) that lacks the type three secretion system to activate PTI. We found that plants infected with different strains displayed dynamic differences in the accumulation of the defense signaling molecule salicylic acid, expression of the defense marker gene PR1, cell death formation, and accumulation/localization of the reactive oxygen species, H2O2. The differences between PTI, ETS, and ETI are dependent on the doses of the strains used. These data support the quantitative nature of PTI, ETS, and ETI and they also reveal qualitative differences between PTI, ETS, and ETI. Interestingly, we observed the induction of large cells in the infected leaves, most obviously with HrcC(-) at later infection stages. The enlarged cells have increased DNA content, suggesting a possible activation of endoreplication. Consistent with strong induction of abnormal cell growth by HrcC(-), we found that the PTI elicitor flg22 also activates abnormal cell growth, depending on a functional flg22-receptor FLS2. Thus, our study has revealed a comprehensive picture of dynamic changes of defense phenotypes and cell fate determination during Arabidopsis-P. syringae interactions, contributing to a better understanding of plant defense mechanisms.
Conflict of interest statement
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

